21 December 2020
The uncharted molecular language of the brain
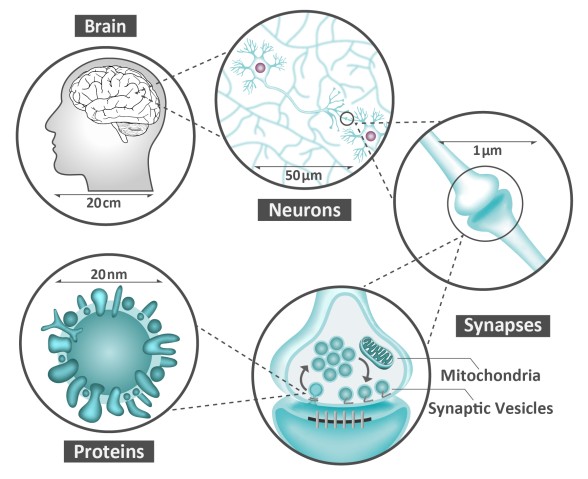
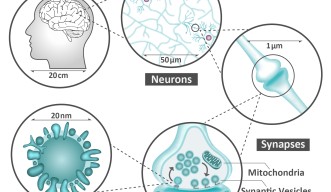
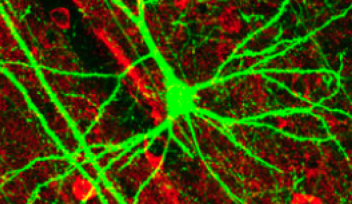
Neurons are like the electrical wiring of our brains, responsible for receiving information from the outside world and conveying this information to the rest of our body. To work correctly, they need to ‘speak’ to each other, and they do this via synapses – specialized structures that act as junctions between neurons. Synapses not only connect neurons but also receive, process, store, and control all the information that flows within this network. Thus, they are fundamentally important for how we operate. A failure within the synapses can influence our memory, spatial orientation, learning ability, and attention span. This failure is also at the root of many brain diseases, such as Alzheimer’s, dementia, autism, ADHD, Parkinson’s, epilepsy and schizophrenia.
Our current knowledge of the full molecular basis of synapses is lacking but a new study, published in PNAS, will help change that. This study has created the most complete annotated resource of the proteins present in the synapses, which could be useful in future health research, such as for providing an earlier diagnosis for brain diseases and identifying more specific drug targets.
“Synapses are full of protein machineries and understanding the contents of these gives us access to so much molecular and functional information,” explained Dr. Zacharie Taoufiq, staff scientist in the Cellular and Molecular Synaptic Function Unit at the Okinawa Institute of Science and Technology Graduate University (OIST) and lead author of the paper. “We knew there were significant gaps in our current knowledge of the synaptic proteome, so we developed a method to look for all the known missing proteins. To our surprise, we found many novel and previously hidden synaptic proteins.”
Involving collaborators from OIST, Max Planck Institute for Biophysical Chemistry in Gottingen, Germany, and Doshisha University in Kyoto, Japan, the research group took a conventional ‘proteomic’ method in this field, which scientists use when they want to identify every protein in a mixture, and modified it to make it more powerful. This new method revealed many hidden sequences of peptides – the building blocks of proteins. Particularly, the group wanted to be able to identify proteins that might largely resemble other proteins but have very different functions.
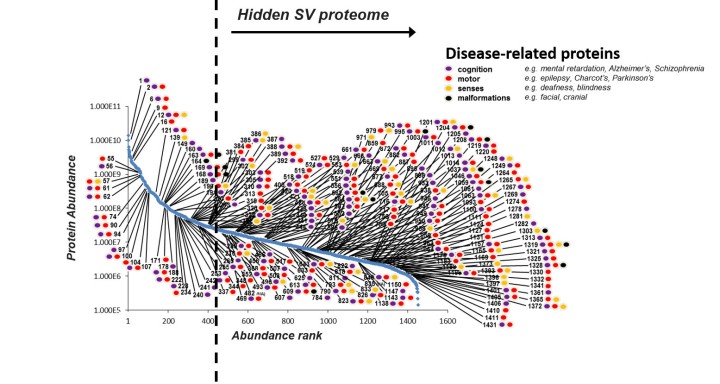
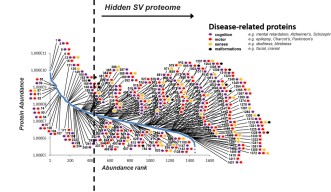
The results were astonishing. In total, the group identified 4439 synaptic proteins of which 1466 were found in synaptic vesicles (SV) – three times more than had previously been known. The research group decided to take a closer look at the SV proteins. They quantified them on a plot, ranking them from most abundant to least abundant. “There was a one-million-fold difference,” said Dr. Taoufiq. “We found a few very abundant ones, which made up 90% of the total amount of SV proteins. But there was also this incredible diversity and what appears to be SV sub-populations. It seems like synaptic proteomes are structured like languages, with a few frequently used words (or proteins) and many less frequent but more specific and meaningful ones.”
To reveal some of the hidden proteins’ functions, the researchers genetically manipulated the neurons to suppress these proteins. One example was a protein that was needed for recycling the vesicle compartment within the synapses. Without this protein, the synapses ability to transmit information decreased in performance. Interestingly, the researchers found that often the ones of lower abundance had some of the most important functions.
“Out of the 1466 SV proteins, we found a link to 200 distinct brain diseases,” Dr. Taoufiq said. “What I found very surprising and interesting is that most of the causes of diseases are linked to the least abundant and previously hidden proteins.”
One of these diseases is Alzheimer’s. The clinical trials for the treatment of Alzheimer’s currently have a failure rate of 99.6%. Dr. Taoufiq theorizes that this is due to patients having symptoms that might appear very similar but are actually caused by different proteins malfunctioning. “This research has resulted in a catalogue of all the different proteins in synapses. This will be a great base for studying synaptic regional and evolutionary diversity of the brain. Our new proteomic method will also be key in finding the molecular cause for disease of each patient. The next difficult but inevitable task.”
Alongside Dr. Taoufiq, this research also included Dr. Momchil Ninov and Professor Reinhard Jahn from Max Planck Institute for Biophysical Chemistry, Professor Tomoyuki Takahashi, Mr. Alejandro Villar Briones, Dr Michael C. Roy, Dr. Han-Ying Wang, Mr. Toshio Sasaki, and Mr. Francois Beauchain from OIST, and Associate Professor Yasunori Mori, Mr. Tomofumi Yoshida, and Professor Shigeo Takamori from Doshisha University.
Specialties
Research Unit
Submit a press inquiry