29 August 2013
Discovery of a New Mechanism Regulating Information in the Brain
When an electrical impulse is transmitted between neurons in the brain to transfer information, a variety of molecules are involved in maintaining the signal transmission. Zacharie Taoufiq and Kohgaku Eguchi, researchers in the Cellular and Molecular Synaptic Function Unit led by Prof. Tomoyuki Takahashi, combined their expertise in biochemistry and electrophysiology and discovered that the enzyme called Rho-kinase plays a critical role in the regulation of signal transmission between neurons.
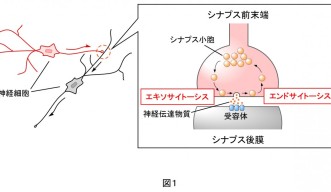
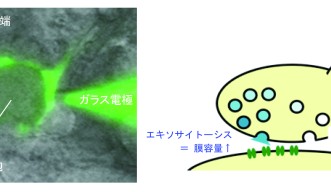
When you take a close look at the junction between neurons, there is a site called the synapse for transferring neurotransmitters from one neuron to another (Fig. 1). The neurotransmitter-releasing side is called the presynaptic terminal and the receiving site is the postsynaptic membrane. The presynaptic terminal contains neurotransmitters enclosed in small spherical intramembrane structures, with a diameter of 50 nm, called synaptic vesicles. When an electrical impulse reaches the presynaptic terminal, the synaptic vesicles fuse with the membrane of the presynaptic terminal and the neurotransmitters are released to the exterior in a process known as exocytosis. The postsynaptic membrane receives the neurotransmitters with its receptors, converts them into electrical impulses, and the information is transferred. The fused membranes of the synaptic vesicles are retrieved in the presynaptic terminals in a procedure called endocytosis, and are later recycled as synaptic vesicles again.
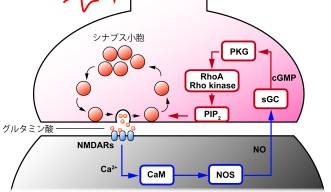
In this research, Dr. Taoufiq and Dr. Eguchi focused on identifying a mysterious molecule that is involved in this endocytosis. Last year, Dr. Eguchi found that the endocytosis is accelerated by nitric oxide released from the postsynaptic membrane and that two chemicals - the enzyme cGMP-dependent protein kinase (PKG) and the lipid phosphatidylinositol-4,5-bisphophate (PIP2) - are involved in this process (Fig. 2) (Eguchi et al., Neuron 2012). However, the mysterious molecule that links PKG and PIP2 remained unclear, and the goal of the research project was to reveal its identity and behavior. “It had been challenging to discover this molecule only by the electrophysiological method,” Dr. Eguchi says. This study progressed significantly after Dr. Taoufiq, who specializes in biochemistry, joined the research.
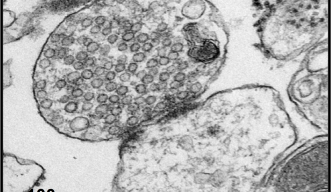
Dr. Taoufiq focused on Rho-kinase, an enzyme that was considered to be the strongest candidate among many molecules. A fraction containing only synapses was isolated from a rat brain sample (Fig. 3) and the number of PIP2 molecules in the fraction was quantified by mass spectrometry (phospholipidomics analysis). They confirmed two things: when a substance that inhibits Rho-kinase activity (inhibitor) was added, the PIP2 content decreased, and when a substance that activates Rho-kinase (activator) was added, the PIP2 content increased. The previous report demonstrated that the PKG inhibitor also decreases the PIP2 content, and the current study demonstrated that the Rho-kinase activator cancels this effect, so the researchers have successfully determined that Rho-kinase is a molecule that links PKG and PIP2.
However, this method itself does not provide an answer concerning when and where the reaction occurs and how it affects neurotransmission. Dr. Eguchi used an electrophysiological method called membrane capacitance measurements (Fig. 4) to investigate the potential difference in the rate of endocytosis that occurs at the presynaptic terminal, when Rho-kinase activity is either inhibited or activated. The result indicated that inhibition of Rho-kinase by the inhibitor indeed decreased the rate of endocytosis. Furthermore, the deceleration of endocytosis caused by a PKG inhibitor is canceled by the Rho-kinase activator, revealing that Rho-kinase links PKG and PIP2 and that it regulates the endocytosis of synaptic vesicles.
“It is rare to see such a thorough study,” Prof. Takahashi says, emphasizing the conclusive result obtained by combining biochemical and electrophysiological techniques. He added that in many studies, even if the presence of certain molecules is known, their behavior in time and space often remains unclear. The methods applied in this study dramatically raised neurotransmission research to a new level where a molecule and a phenomenon could be linked to each other. There are high expectations that these methods will facilitate an understanding of neurotransmission-related diseases such as Parkinson’s and Alzheimer’s to provide both better treatment and diagnosis in the future.
The result of this study was published in the July 17, 2013 edition of the Journal of Neuroscience.
Specialty
Research Unit
Submit a press inquiry