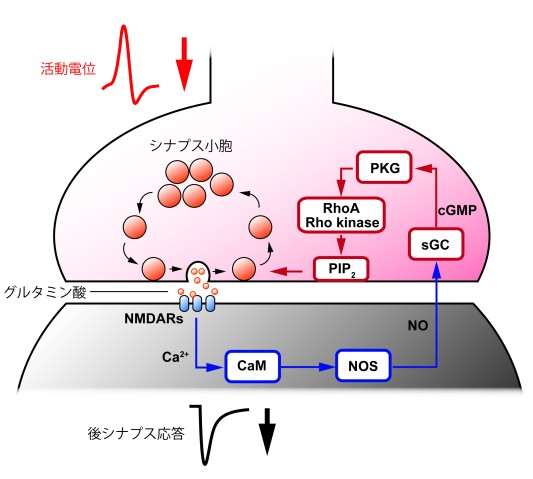
Retrograde regulation of vesicle endocytosis via RhoA/Rho-kinase
Zacharie Taoufiq and Kohgaku Eguchi, researchers at the Cellular and Molecular Synaptic Function Unit led by Prof. Tomoyuki Takahashi, combined their expertise in biochemistry and electrophysiology and discovered that the enzyme called Rho-kinase plays a critical role in the regulation of signal transmission between neurons.
Neuronal information is usually passed anterogradely from presynaptic cells to postsynaptic cells, but there are retrograde signals conveyed from postsynaptic cells to presynaptic terminals. Nitric oxide (NO) is one such signal molecule. It regulates vesicle endocytosis in the presynaptic terminal. After the neurotransmitter glutamate is released from the presynaptic terminal, it binds to postsynaptic NMDA receptors and induces calcium ion (Ca2+) influx into the postsynaptic cell. The high calcium concentration activates calmodulin (CaM), thereby promoting NO synthesis by activating NO synthases (NOS). NO is diffused into presynaptic membranes and activates soluble guanylate cyclase (sGC), which synthesizes cyclic GMP (cGMP) from GTP. Increasing cGMP activates cGMP-dependent protein kinase (PKG), thereby increasing PIP2 concentration in the presynaptic terminals and then accelerating vesicle endocytosis. This study showed that a small GTPase RhoA and the associated protein kinase (Rho-kinase) intermediated between PKG and PIP2 in the retrograde regulation system of vesicle endocytosis.
The result of this study was published in the July 17, 2013 edition of the Journal of Neuroscience.
Copyright OIST (Okinawa Institute of Science and Technology Graduate University, 沖縄科学技術大学院大学). Creative Commons Attribution 4.0 International License (CC BY 4.0).
Tags