26 August 2019
Advanced Biosensor Shines Light on DNA Sequencing
DNA sequencing technologies are indispensable in medicine, although having quick access to results has historically been a challenge. As part of a collaboration, researchers at the Okinawa Institute of Science and Technology Graduate University (OIST) have developed a new technique that monitors how DNA interacts with enzymes in real time — which could find use in sequencing applications.
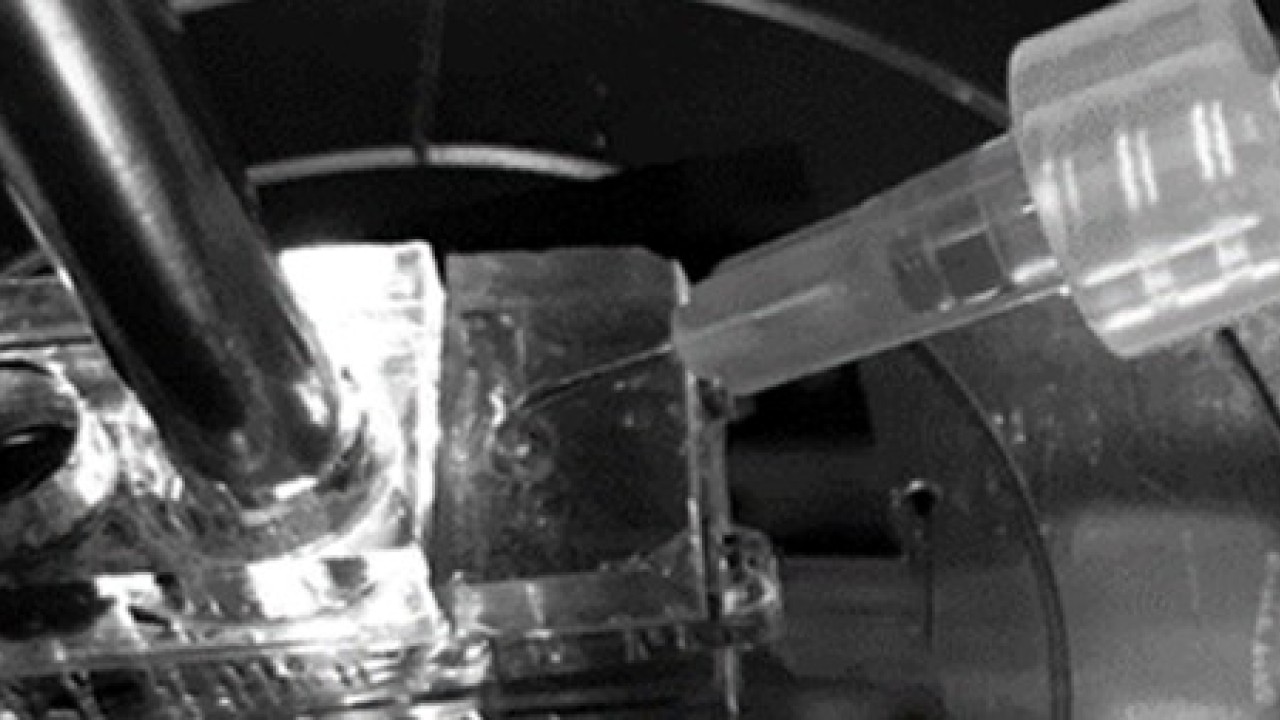
Writing in Biosensors and Bioelectronics, the team explains their proof-of-concept biosensor that uses a microfluidic chip with light sensing to provide researchers with real-time readouts of a reaction. The reaction takes place on the chip and is detected by the light sensor. This sensor eliminates the need for labels — speeding up the process — meanwhile the tiny chip improves the biosensor’s sensitivity.
“Building this biosensor has involved experts in microfluidics, DNA and enzymes, and nanomanufacturing,” says Dr. Nikhil Bhalla, who worked on the paper as a postdoctoral scholar in OIST’s Micro/Bio/Nanofluidics Unit. “That it allows for such collaborations is what makes OIST so special.”
“We will now work on the device’s sensitivity, throughput, and potential use in biotechnology.”
Lab-on-a-chip sequencing
Understanding how DNA interacts with enzymes as a reaction happens gives researchers detailed information — which is more useful than simply knowing the outcome of a reaction after the fact. Until recently, studying such interactions has often relied on the use of labels. This slows down the process, and the labels can be damaging to the compounds being studied.
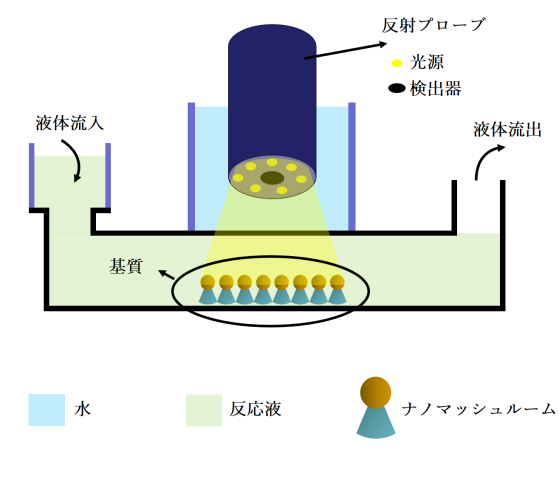
Localized surface plasmon resonance (LSPR) has emerged as a label-free alternative that uses light sensing in place of probe chemicals. In this approach, a small device shines a light onto the reactants and is able to detect small shifts in light absorbance that correspond to a reaction taking place. The device then feeds live results to a screen.
The team combined LSPR with microfluidics, placing reactants on a tiny plastic chip that is loaded into the LSPR device. In this study, the researchers tested the approach on DNA replication, an important reaction that creates a copy of our DNA every time our cells divide. The chip contains an array of mushroom-shaped structures, to which the reactant — DNA in this example — can stick while an enzyme does its work.
As the enzyme binds to the DNA and creates an extra copy, the device detects a change in light absorbance. This shows that a reaction is taking place. The changing light absorbance is then displayed on an attached screen that updates in real-time as the biosensor’s readout.
Having access to sensitive devices offering instant readouts is invaluable in many contexts. Lab-on-a-chip devices, like this example, are especially helpful when trying to understand a patient’s genetic risk for disease. Instead of sending samples to the lab and waiting days for results, fast and accurate genetic testing can take place with the patient — and can be immediately used in decision-making.
And beyond patient settings, quick and easy-to-use diagnostics are also needed in clinical, pharmaceutical, and scientific research, as well as forensic science. The OIST team’s biosensor illustrates what is, and what might be, possible in the world of real-time monitoring. In the future, this could extend beyond DNA replication to other processes like DNA sequencing.
For now, the team will work on enhancing the sensitivity of the device by understanding the optimal ratio of reactants placed on the chip. They also intend to build additional features, such as temperature control, into the device. These improvements will support the team’s parallel efforts in point-of-care testing — bringing new assets to patients and healthcare professionals.
This paper was a collaboration between researchers at the Okinawa Institute of Science and Technology Graduate University (OIST) and the Karlsruhe Institute of Technology (KIT).
Research Unit
Submit a press inquiry