19 May 2011
Two-photon Microscopy: A powerful tool to observe neuronal activity
|
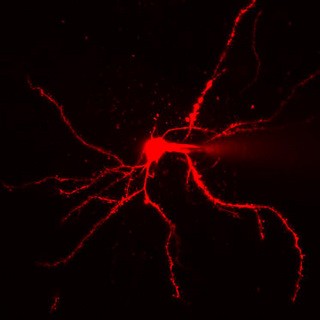
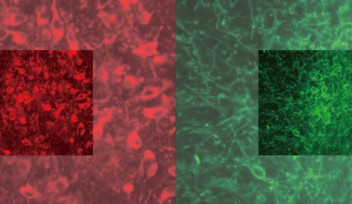
A two-photon microscopic image of a neuron fluoresced with red dye |
By Kaoru Natori
What do you do if you want to observe a tiny object? You could use a magnifying glass. But what if the object is even too small to see through the magnifier and has the additional complication of being inside a living organism, for example in the brain of a mouse?
Researchers at OIST are interested in the basic units of which our brains are composed - so called nerve cells or neurons. These cells have a diameter of about 10 microns (.01 mm) with processes that can reach even more than one meter but are thinner than 1 micron (0.001 mm). Billions of neurons, each with tens of thousands of connections to others, engage in non-stop, busy electrochemical conversations. The signals they send result in our thoughts, actions, words and emotions. However, how exactly behavior arises from cellular activity and how information is processed in the brain remains a mystery.
To study the brain on a cellular level, scientists have developed highly sophisticated microscopy techniques. On one hand, neuronal activity has to be made visible and on the other hand, microscopes are needed that can detect these signals.
When special fluorescent dyes are injected into a living tissue, the neurons not only get labeled and so visible when excited by light of the right wavelength, but the activity of the labeled neurons also changes the fluorescence of the dye molecules. As a result, the neuronal activity is transformed into an optical signal. By measuring the optical signal, the cellular activity in a living tissue can be studied in real time and almost simultaneously from different locations.
To record these dye signals, one of the most advanced microscopy techniques is used, called two-photon laser scanning microscopy. It is a fluorescence imaging technique that relies on the near-simultaneous absorption of two photons by a molecule to excite fluorescent dyes. A two-photon microscope uses an infrared laser beam that is focused through an objective lens and excites dye molecules only within a tiny focal spot less than 1 micron in diameter. The microscope can then move the focal spot through the sample, and so scan the tissue with great accuracy.
|
A two-photon microscopy |
Two-photon laser scanning microscopy permits three-dimensional tissue imaging of up to 1 mm depth. While this may not sound like a lot, it really is for many applications in biology. At OIST, researchers use this technique to study several different questions about the brain and its function.
Studying Parkinson’s disease
For communication between neurons to occur, an electrical impulse travels down the tail of a cell, called the axon, and arrives at the junction of two neurons known as a synapse. The impulse is then transmitted via chemical signals onto a dendrite of another cell at the synaptic gap. In people with Parkinson’s disease, the transmission of a chemical signal, carried out by neurotransmitters stored in synaptic vesicles of an axon, is hindered. Dr. Takuya Hikima of the Brain Mechanism for Behavior Unit uses a two-photon microscope to image how synapses work. More specifically, he is trying to quantify vesicular release from cortical cell axons in the cerebrum. His aim is to develop a concrete method to visualize synaptic vesicle dynamics in Parkinson’s disease.
|
Dr. Hikima using a two-photon microscope |
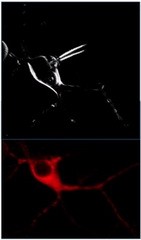
Simultaneous views of the same cell. In the top, only the cell's outer membrane is visualized while the bottom one shows the whole neuron filled with red dye. |
Neuronal activity is indicated by membrane voltage changes in neurons followed in many cases by calcium concentration changes. Studies have shown that dysfunction of the basal ganglia, located deep within the brain, contributes to major neurological and behavioral disorders including Parkinson’s disease, in which dopamine-producing cells die off. Using a two-photon microscope with transgenic mice that express fluorescent markers in dopamine receptors, Dr. Tomomi Shindou of the Neurobiology Research Unit examines the striatum of the basal ganglia. He is employing the two-photon microscopy and electrophysiological techniques called patch-clamp recordings to measure calcium concentration and changes in cell membrane potential at the same time. His goal is to deepen understanding of events taking place at individual dendritic spines, which are small door knob-shaped extensions from a neuron's dendrite that receives input from a single synapse of an axon.
|
Patch clamp system with two-photon microscopy |
Dr. Shindou performs imaging of a brain slice from a transgenic mouse |
Brain imaging in the living animal
While the above described questions can be studied in brain slices, Dr. Bernd Kuhn, who heads the Optical Neuroimaging Unit at OIST, is aiming to study neurons in the brain of living mice. Of course, animal welfare has highest priority in such studies and is at the same time crucial for high quality scientific results. For this very special application, Dr. Bernd Kuhn is building a special two-photon laser scanning microscope. The customized device, which will be in use as early as this fall, enables real-time monitoring of the neural network activity of a behaving animal on different levels of brain organization – the activity in the local neuronal network, a single neuron, and even parts of a single neuron and everything within the living animal!
|
Two-photon microscope with spherical treadmill will be used for brain imaging in living mice |
Development of new techniques has allowed us to have a much closer look at events taking place in the brain. Better understanding of the activity holds a key to answer the questions of behavior arousal and information processing. OIST researchers are trying not only to deepen the understanding of the brain, but also to go beyond the current technical limitations. The tools being developed at OIST will be applicable to many different fields in neuroscience, and lead to better treatments of neurological disorders.
Specialty
Research Unit
Submit a press inquiry