28 February 2019
Too-Tight Membrane Keeps Cells from Splitting
Cells divide to grow new tissues or patch up damaged ones, but when cell division goes wrong, it can cause more harm than good. To avoid dire consequences, namely disease and unwanted cell death, cells employ a suite of failsafes to ensure they split evenly, every time. Now, scientists have identified a previously unknown mechanism by which cells accomplish this impressive feat.
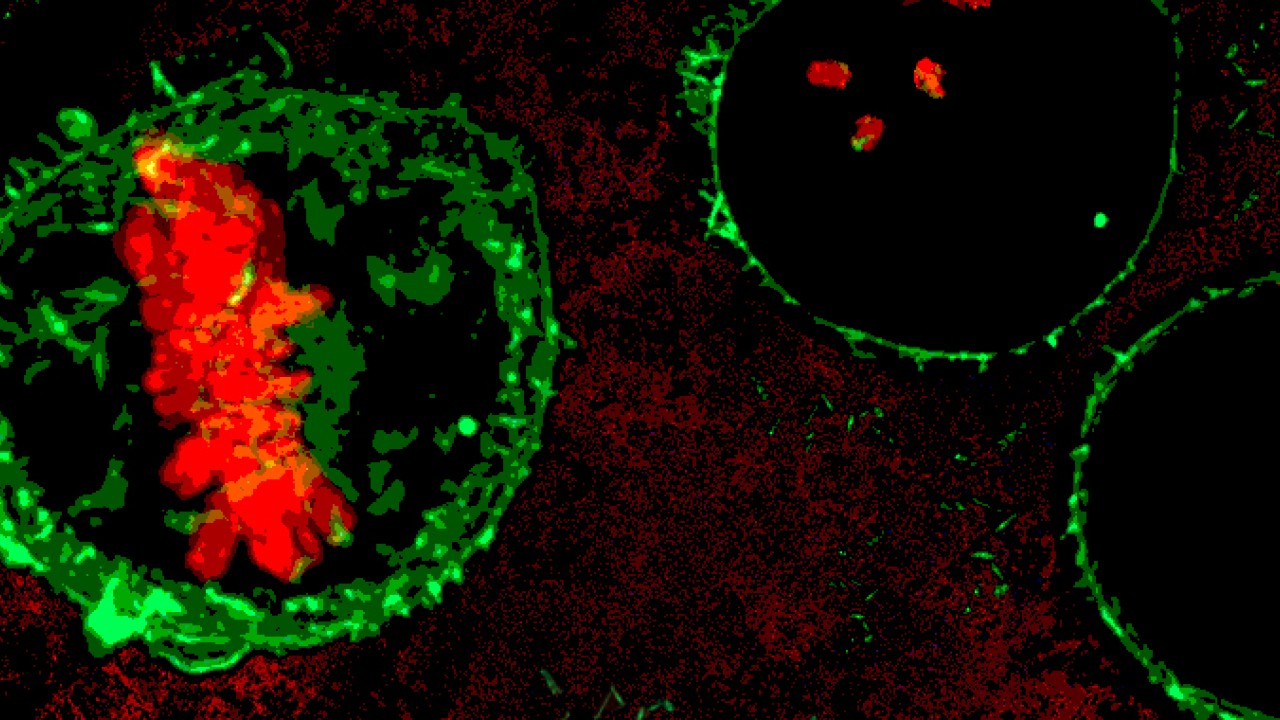
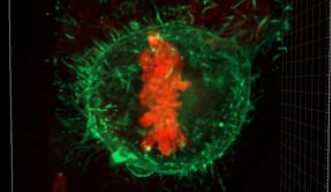
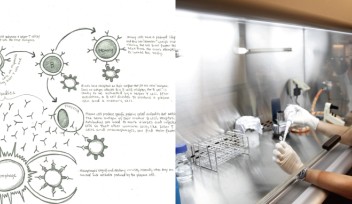
Usually, the genetic material within a cell assembles in loose, lengthy strands—picture an unwound spool of thread. During cell division, those strands coil themselves into tightly bound chromosomes and align at the cell’s equator. To make room for this chorus line formation, the squishy cell rounds up into a perfect sphere. Scientists have long known that if a cell cannot round at this stage, it cannot divide. The new research, published February 28, 2019 in Nature Communications, presents a novel idea: if a cell rounds with too much force, division becomes equally impossible.
“There are many reported cases of cells not having enough force to round up, but this is the first case reporting excess force,” said Prof. Keiko Kono, co-senior author of the study and principal investigator of the OIST Membranology Unit. The researchers discovered an alternate scenario, wherein a critical failsafe is removed, the cell generates too much force and division simply stalls. Though chromosomes still organize along the cell equator, a stage known as the “spindle assembly checkpoint,” division cannot proceed past this point.
“The chromosomes are just beautifully aligned,” said Kono, “but the checkpoint cannot be released.”
Kono conducted the study with co-senior author Prof. Makoto Nakanishi of the University of Tokyo, as well as additional collaborators from the University of Tokyo, Yamaguchi University, Nagoya City University, Kyoto University, and the Japanese Foundation for Cancer Research. At OIST, the Kono Unit pursues related research, investigating how cells change shape in response to different stimuli, including damage to their outer membranes. The research advances our understanding of how cells work and could someday inform how we treat many medical ailments, from hearing loss to cancer.
Molecular Brake Helps Maintain Balance
In a petri dish, cells tend to lie flat like fresh baked cookies stuck to a baking tray. During division, they puff up on the dish, forming into tiny balls. It’s been shown that a protein called cyclin-dependent kinase 1, or Cdk1, helps drive this rounding action. Cdk1 triggers a chain reaction which leads to accumulation of stringy filaments, called F-actin, at the cell surface. F-actin works with a proteinous partner—myosin—to pull the surface taut and cause the cell to round.
“But there’s a kind of puzzle here,” said Kono. At the onset of cell division, tension at the cell surface steadily increases. But once chromosomes align at the equator, the tension plateaus and remains nearly constant until the cell finally splits. “[The established theory] explains this linear increase, but it doesn’t explain why the force is stabilized—our work explained this.”
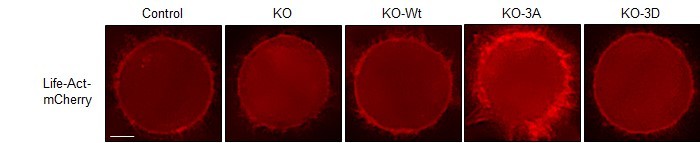
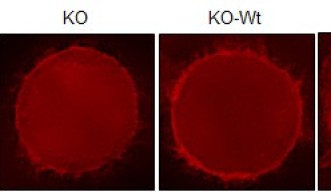
To solve this mystery, the researchers created mutant cells in which the chain reaction set off by Cdk1 was disrupted. Normally, Cdk1 triggers a series of events that cues the cell to build actin filaments. Acting like a key in the ignition, a protein downstream of Cdk1 activates the F-actin assembly line, stringing together molecular links to create each individual filament. The scientists suspected that, if one protein jumpstarts this process, another must act as a brake to slow it down.
They found that, remarkably, Cdk1 steps in to play this role. After cueing the cell to construct actin filaments, Cdk1 modifies the assembly line itself, halting construction before too much actin accumulates. In mutated cells, in which Cdk1 was prevented from putting on the brakes, actin piled up at the cell surface and caused tension to build uncontrollably. Unless thebrake was reinstalled, the mutant cells could not divide.
‘Goldilocks Zone’ for Cell Division
The results suggest that excess tension at the cell surface disrupts division, but further research is needed to know exactly why. In the future, Kono plans to investigate how forces acting at the cell membrane affect the timing of division, especially on either side of the spindle assembly checkpoint. Following the checkpoint, the perfect row of chromosomes gets torn in two, granting an equal amount of genetic material to each resulting daughter cell.
In preparation for this critical moment, the cell needs everything to go just right.
“If the chromosomes cannot be aligned and distributed well, it will immediately induce diseases,” said Kono. For chromosomes to be divvied up evenly, the cell must round up just enough, but not too much. “It’s intuitive to imagine if you have less force, you can’t round up. But if you round up too much, this is also harmful; this is a new idea.”
Specialties
Research Unit
Submit a press inquiry