20 May 2016
Tampering with the Current in a Petri Dish
Electricity plays a key role in cell studies, but practical issues linked with the shape of the laboratory cultureware have troubled this research. Laboratory cultureware are the plastic containers used by researchers to grow cells. These containers are typically shallow cylinders: a classic example is a petri dish. While a petri dish is circular, the simplest way to create a uniform electric field is based on a rectangular shape. These different geometries prevent scientists to fully exploit the potential of a cell cultureware, as a significant part of the round petri dish base remains outside the field-generating rectangle that goes into the cultureware. A PhD student’s project, which has led to a patent application and a published article in Scientific Reports, is radically changing this situation.
The student, Hsieh-Fu Tsai, worked under the supervision of Prof Amy Shen, head of the Micro/Bio/Nanofluidics Unit at the Okinawa Institute of Science and Technology Graduate University (OIST). His research project focused on cell behaviour in an electric field. “Cells respond to electric current,” Tsai explained. “Some cells migrate towards the positive pole, while others towards the negative pole, and some cells show a specific alignment with the electric field.” These phenomena are known to play an important role in key biomedical areas, like wound healing and the early stages of cell development, such as neurogenesis and embryogenesis.
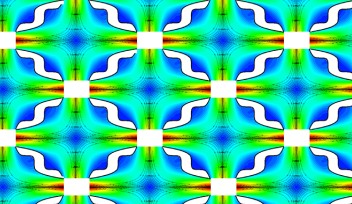
Scientists typically choose to study cells in a uniform electric field, as such an even field is the simplest case to work with in a controlled setting. An effective way to create a uniform electrical field is through a rectangular device, because the electric poles are connected to two of the opposite sides of the rectangle, and thus the pathways of the electric current are all of the same length.
However, most standard cell cultures happen in a circular shaped environment, like a petri dish, and it is not possible to directly create a uniform electric field just connecting electric poles to the opposite sides of a circle. “The walls of a petri dish are curved,” Prof Shen commented, “and in a circle the pathways of the electric current are of different lengths, so the resulting electric field is not uniform.”
Tsai and colleagues found a revolutionary solution to this problem. They created a plastic insert that modifies the pathways of the electric current in a circular shape, making each current path of the same distance. The insert, simple and inexpensive, achieves this goal by making the shorter pathways running inside the device itself, and thus extending their length until they match the longer pathways.
The insert has four holes on top: two holes for providing cells with nutrient, and two holes for applying electricity. First, the researchers lay cells on the bottom of a petri dish. Then, the insert is placed into the cultureware and sealed in place. Finally, the scientists add fresh nutrient for the cells to grow and apply the electrical current.
The design of the insert is based on the fundamental principles of electricity, which helped the researchers in finding the optimal shape of the device. Once the shape is defined, the insert can be directly created with a 3D printer. Thanks to this simple process, the insert is scalable and can be easily adapted to fit most of the common laboratory cultureware of any size. OIST scientists have already tested the performance of the device through a successful experiment on mouse embryonic fibroblast cells.
“One of the advantages is that, with this device, researchers can use most of the surface coverage of the dish,” Prof Shen said. “This results in higher cell count, and thus in more samples for further experiments.”
There are several applications for this device in cells studies. “This time we were specifically aiming for tissue applications, because many researchers are trying to create functional body tissues in the lab; for example, muscle, skin, and liver,” Tsai explained. “You can grow these tissues, but frequently they do not have the function that you see in the body. That’s because they are not mature yet: they need training, like a muscle needs exercise. An electric field is one of the training method scientists are trying to use on cells.” The project has already generated contacts with the tissue engineering industry.
Notably, Tsai developed this project during a three-month lab rotation that is part of the standard curriculum at OIST. PhD students work in three different laboratories during their first year, exploring the diversity of scientific research. This model, possibly unique in the tertiary education landscape, is proving itself successful and effective in fostering innovation.
The research was interdisciplinary and done in collaboration with Prof Tadashi Yamamoto, leader of OIST Cell Signal Unit, and Dr Ji-Yen Cheng’s group from Academia Sinica in Taiwan.
Specialties
Research Unit
Submit a press inquiry