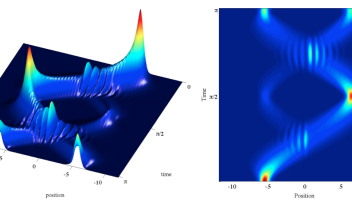
The Light-Matter Interactions Unit

The Light-Matter Interactions Unit
Date:
30 November 2012
Copyright OIST (Okinawa Institute of Science and Technology Graduate University, 沖縄科学技術大学院大学). Creative Commons Attribution 4.0 International License (CC BY 4.0).
Tags
Faculty
Researchers
Staff
Students