28 September 2022
New model captures the erratic speed of DNA copying proteins in bacteria
Cell division is fundamental for life, allowing organisms to grow, repair tissues, and reproduce. For a cell to divide, all the DNA inside the cell (the genome) must first be copied, in a process called DNA replication. But the precise dynamics of replisomes – the protein machinery that copies DNA – has been difficult for scientists to determine.
Now, researchers at the Okinawa Institute of Science and Technology (OIST) in Japan have developed a new model that can determine variations in the speed at which replisomes copy bacterial genomes. The model, combined with experiments, shows that certain sections of DNA are copied faster than others and reveals an intriguing link between replication speed and error rate. The research was published in eLife on July 25, 2022.
"The machines that copy DNA are amazing – they are very fast and very precise,” said Simone Pigolotti, an Associate Professor at OIST who heads the Biological Complexity Unit. “Understanding these machines can tell us what is important for cells – what mistakes are tolerable, what mistakes are not, how fast replication should be.”
The model relies on measuring how abundant different DNA locations are within a population of bacterial cells that are constantly dividing. In bacteria, to start DNA replication, two replisomes attach to the DNA at a set origin point and head in opposite directions along the loop of DNA, copying DNA until they meet on the other side. This means that the DNA closest to the origin point is copied first, while DNA closest to the termination point is copied last.
“If you let a population of bacteria freely grow, then at any given point in time, most cells will be in the process of cell division. Because DNA replication always starts from the same location, this means that if you then sequence all the DNA, there will be a higher abundance of DNA that is closest to the origin point, and a much lower amount of DNA that is closer to the end point,” explained Prof. Pigolotti.
In the study, researchers from the Nucleic Acid Chemistry and Engineering Unit at OIST cultured Escherichia coli (E. coli) bacteria at different temperatures. The Sequencing Section then sequenced the bacteria’s DNA.
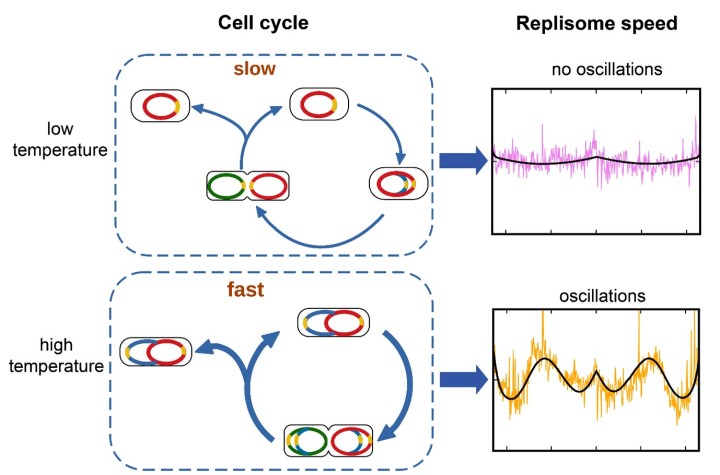
By analyzing features of the distribution curve, the researchers were able to determine the exact speed of the protein machinery. They found that as the temperature increased, the replication speed increased. Even more interestingly, the researchers discovered that the replisomes varied their speed at different points along the genome.
One potential reason for their fluctuating speed, Prof. Pigolotti speculates, is that there may be limits on resources needed for replication, such as nucleotides – the building blocks of DNA.
In E. coli, when conditions are good, a single bacterial cell can divide every 25 minutes. But the process of replicating DNA takes longer – around 40 minutes. Therefore, in order to keep up at high growth rates, multiple copies of the genome are replicated at the same time, which increases the number of replisomes at work. Competition for nucleotides could then cause the replisomes to slow down.
Additional evidence backs up this hypothesis. At low temperatures and in nutrient-poor cultures, when the growth rate of the bacteria is low and only one genome would be copied at a time, these fluctuations in replication speed disappear.
Intriguingly, the researchers also found that the oscillations seen for replication speed also matched the oscillations in mutation rate documented in other studies. When they overlaid the two patterns, they found that areas of the genome that were copied faster also had a higher mutation rate.
"This seems intuitive – if we think of an action, like typing on a keyboard, the faster we type, the more likely that we will make a mistake,” said Prof. Pigolotti. “So we think that when the replisomes go faster, their error rate is higher.”
For Prof. Pigolotti, the next step is to determine how the speed of replication changes in mutant strains of E. coli, such as ones that are missing proteins that assist in replication. He is also curious to see if the pattern holds in other strains of bacteria.
"It’s a really exciting research direction,” said Prof. Pigolotti. “And all the work was done in collaboration with other units here. It’s the kind of interdisciplinary collaboration that can only happen at OIST.”
Article Information
Specialties
Research Unit
Submit a press inquiry