07 May 2021
OIST Professor Mahesh Bandi engineers DIY oxygen concentrator
As cases of COVID-19 in India surge to unprecedented highs, there have been fatal shortages in medical-grade oxygen. Professor Mahesh Bandi, down on the ground in Hyderabad, India, has now engineered two devices capable of producing medical-grade oxygen.
Instructions for building the device have now been published on his website.
Medical-grade oxygen, classified as a gas mixture where oxygen concentrations are 95% or higher, is a life-saving treatment used for patients with severe COVID-19 who are experiencing difficulties with breathing. In India, where supply has lagged perilously behind demand, prices for oxygen tanks have soared to up to ten times the market price on the black market.
“The situation in India is like something I’ve never seen before,” said Professor Mahesh Bandi. “It’s hard to believe it’s happening in the 21st century. Everyone knows someone who has died of COVID-19 or has friends and relatives in critical need of oxygen.”
Working to alleviate this crisis, Prof Bandi, who leads the Nonlinear and Non-equilibrium Physics Unit at OIST, has used locally sourced materials to design and build two devices. The larger device is capable of producing just over 100 liters of medical-grade oxygen per minute, while the smaller, portable device can supply 9 liters per minute. A single patient with severe COVID-19 may need around 5-15 liters of oxygen every minute.
Instead of producing oxygen through chemical reactions, the device simply concentrates the oxygen found in normal air within the atmosphere. The air we breathe is predominantly composed of nitrogen, at around 78.1%, with approximately 21% oxygen and trace amounts of other gases. By processing the air so that the nitrogen is removed, the device can create gas mixtures with oxygen concentrated to around 95%.
“It’s a similar method to one I used back as a PhD student to cheaply create pure oxygen for my experiments,” said Prof. Bandi. “But the biggest challenge this time was to try and build these devices without a lab.”
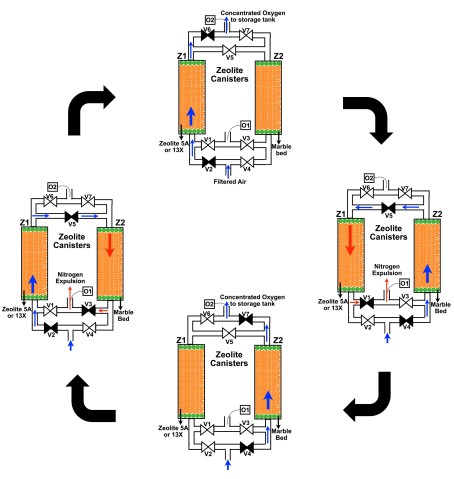
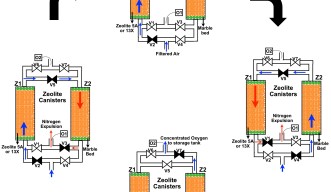
The device works by using an air compressor to create a pressurized flow of air, which is then passed through an air filter made from silica gel. The gel removes impurities and water vapor that would otherwise reduce the efficiency of the device.
Once filtered, the pressurized air enters the nitrogen removal stage, which occurs in a cycle of four standard steps. The flow of air alternates between two different cylinders which contain zeolite – an artificial compound that acts as a molecular sieve, allowing some molecules to pass through, while stopping others. In these devices, Prof. Bandi used Zeolite 5A and 13X, which molecules of nitrogen “stick” to, while oxygen can pass through.
In step one, the air is filtered through the first cylinder and the concentrated oxygen is then stored in an oxygen tank. However, after five minutes, the zeolite in the cylinder is unable to hold any further molecules of nitrogen.
In the second step, the nitrogen stuck to the zeolite is purged from the system. To do this, valves are used to direct the pressurized flow of air through the second cylinder and then back into the first cylinder for 100 seconds. The concentrated oxygen “knocks” the nitrogen away from the zeolite, and the nitrogen is then released into the atmosphere.
In step three, the pressurized air continues to flow through the second cylinder but is now diverted into the oxygen storage tank. After five minutes, the second cylinder can no longer hold anymore nitrogen and so in the fourth step, the air is sent through the first cylinder and back into the second cylinder to purge the nitrogen for 100 seconds.
“The cycle can then be repeated over and over to create a near continuous supply of concentrated oxygen,” said Prof. Bandi. Because the oxygen is then stored in a tank, patients are not affected by the brief cut off in oxygen supply while the nitrogen is being purged.
Currently, the valves used by the device are manual, meaning that someone must continually control the flow of air while the device is running. In the future, Prof. Bandi hopes that automatic valves can be sourced that would prevent human errors and allow the device to be run for longer periods of time.
Prof. Bandi also noted that the device runs more efficiently in cooler temperatures and should be used in well-ventilated areas to prevent a build-up of nitrogen.
Since publishing his instructions, five incubator companies, three non-governmental organizations and six academic teams across India have started building and shipping concentrators to those in desperate need of medical-grade oxygen.
“It’s incredible to hear that people have already started to make their own devices,” said Prof. Bandi. “These devices may seem complicated, but actually they are quite simple and can be the difference between life and death for those with severe COVID-19. Every life saved through these devices that might otherwise have been lost, makes the effort a worthy one.”
###
Header image: A patient, wearing an oxygen mask, sits outside Lok Nayak Jai Prakash Narayan Hospital (LNJP), one of India's largest facilities for coronavirus disease (COVID-19) patients only, in New Delhi, India, April 22, 2021. REUTERS/Adnan Abidi
Research Unit
Submit a press inquiry