12 April 2019
Reactive Research and Modifying Metals
Publishing as first author is a notable feat. Recently, Okinawa Institute of Science and Technology Graduate University (OIST) PhD student Sébastien Lapointe published a paper in Organometallics as first author, making it the first in his PhD. Lapointe is in the Coordination Chemistry and Catalysis Unit led by Prof. Julia Khusnutdinova.
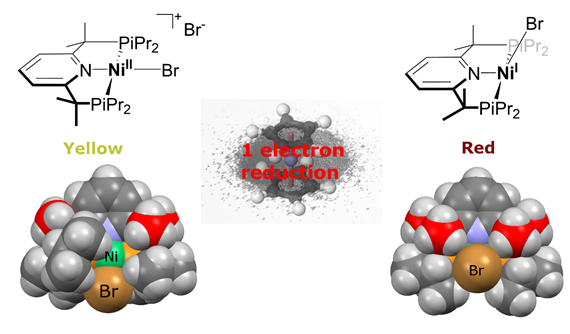
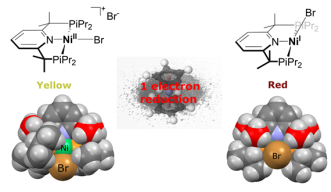
The scientists modified pincer ligands – a molecule that’s a chelating agent – which binds with a metal center. Lapointe wanted to see if he could stabilize reactive complexes with his ligand, and his results from the experiments showed that he could.
Nickel complexes are often catalysts that are used for polymerization and making fine chemicals in industry, but the intermediates in catalysis are poorly understood. This is because one of the things that make nickel complexes so active, unpaired electrons, also make it impossible for chemists to use their most important spectroscopic technique (NMR) to follow along with the reactions and see short-lived intermediates.
“The main factor that determines the stability of our complexes are the presence of the methyl groups on the arm. They have three roles, blocking unwanted reactions on the ligand, increasing the size of the ligand and donating electron density to the metal.” Lapointe said.
The experiment’s results can be visually seen. Follow the photos below to get a taste of the research.
Specialty
Research Unit
Submit a press inquiry