27 November 2018
Signature Structure Grants Enzyme Superfamilies Their Power
Like a bustling factory floor, the inside of a cell requires many workers to keep business running smoothly. Superfamilies of enzymes known as methyltransferases, or MTases, help to turn genes on and off, modify cellular products, and regulate signals cascading through the cell. The structure of an MTase equips it with the right tools to do its job correctly, and understanding these structures allows scientists to optimize those tools, or swap them out for new ones.
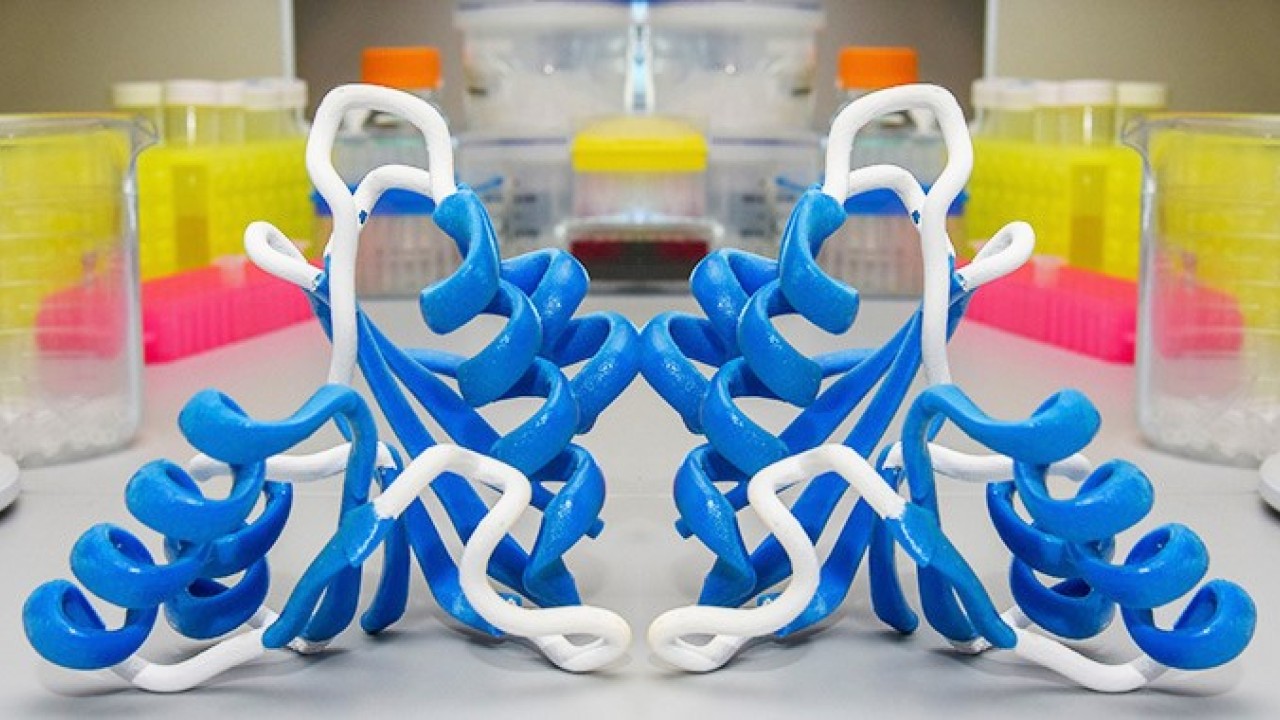
Now, researchers at the Okinawa Institute of Science and Technology Graduate University (OIST) have uncovered a key feature of the largest class of methyltransferases, which are known as Rossmann-fold MTases.
“The Rossmann-fold is very abundant in nature; many proteins have this structure,” said Dr. Bhanu Pratap Singh Chouhan, first author of the study and a postdoctoral scholar in the Protein Engineering and Evolution Unit, led by Prof. Paola Laurino. “But what sets this one apart? That was the question.”
The study results, published in Biochemistry on November 8, 2018, pinpoint a signature structural element in Rossmann MTases that appears critical to their function and is located on a loop near their reaction center.
Loop region takes a turn
“Loops” within proteins connect secondary structures known as α-helices and β-strands and are generally very flexible. Scientists can induce mutations on these loop regions to alter the structure and function of proteins, thereby gaining insights into their mechanisms.
“Generally, loop regions are considered highly evolvable as they are more tolerant of mutations,” said Chouhan. “You can evolve loop regions without completely disrupting the function of a protein.” In the new study, the researchers aimed to learn more about the key structural elements of Rossmann MTases so that scientists could modify these protein components in the future.
Using the publicly-available Protein Data Bank, the researchers analyzed and compared the structures of over 190 Rossmann MTases. They found that over 90 percent of the Rossmann MTases displayed a common structural feature in one specific loop region.
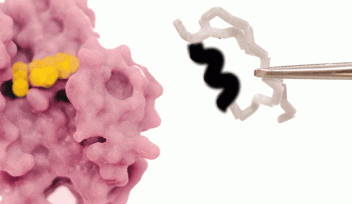
“We were able to show that this loop region adapts a particular 3D orientation and is a key feature of Rossmann MTases,” Chouhan said. Four amino acids make up the marker, known as a type-II’ β-turn conformation, and appear to grant the loop stability. This stability aids the enzyme’s function because the loop neighbors a cofactor – a substance whose presence is essential for the enzyme to work – and its slight rigidity fixes it in place. This precise positioning enables Rossmann MTases to fulfill their primary purpose: chemically modifying DNA, RNA, proteins, small molecules, and lipids in order to tune their function.
Key structural feature is highly conserved
Many enzymes bear the iconic Rossmann-fold, but not all these enzymes are MTases. The researchers compared Rossmann MTases to other Rossmann enzymes and found that this β-turn conformation was significantly more prevalent in Rossmann MTases than their counterparts. The result suggests that the β-turn is specific to Rossman MTases and integral to their function.
To test this theory, the scientists also modeled what would happen if the β-turn became mutated and lost its key structural features. They found that Rossmann MTases seem to lose stability when this β-turn region is altered.
The researchers went on to validate their results experimentally by shuffling chemicals around in actual Rossmann MTases. In the lab, scientists can direct proteins to mutate, evolve and take on new functions. But if they alter an essential element in a protein that is integral to its function, strong selective pressure drives the protein to correct that change. The researchers created a library of about 1 million mutated Rossmann MTases and found that, in just three rounds of evolution, all the modified β-turn conformations returned to their signature structure.
The results suggest that this characteristic of Rossmann MTases is highly conserved and critical to the enzymes’ function. Now that the dots between structure and function are connected, scientists may be able to design new Rossmann MTases more effectively.
“We can basically think of this as providing a template for designing loops for MTases in the future,” said Chouhan.
Specialties
Research Unit
Submit a press inquiry