22 July 2016
The Eyes are the Window into the Brain
Our eyes are constantly moving, whether we notice or not. They jump from one focus point to another and even when we seem to be focused on one point, the eyes continue to reflexively move. These types of eye movements are called saccades. During saccades, even if we are not actively thinking about moving our eyes, our brain is still quickly working to land our focus precisely on spots that hold important information, such as the eyes of a person that you are talking to. The saccade is an example of sensorimotor coordination – how we coordinate our movement with what we sense –and has a far-reaching impact on motor control. Therefore, it is important to understand what is happening in the brain during saccades.
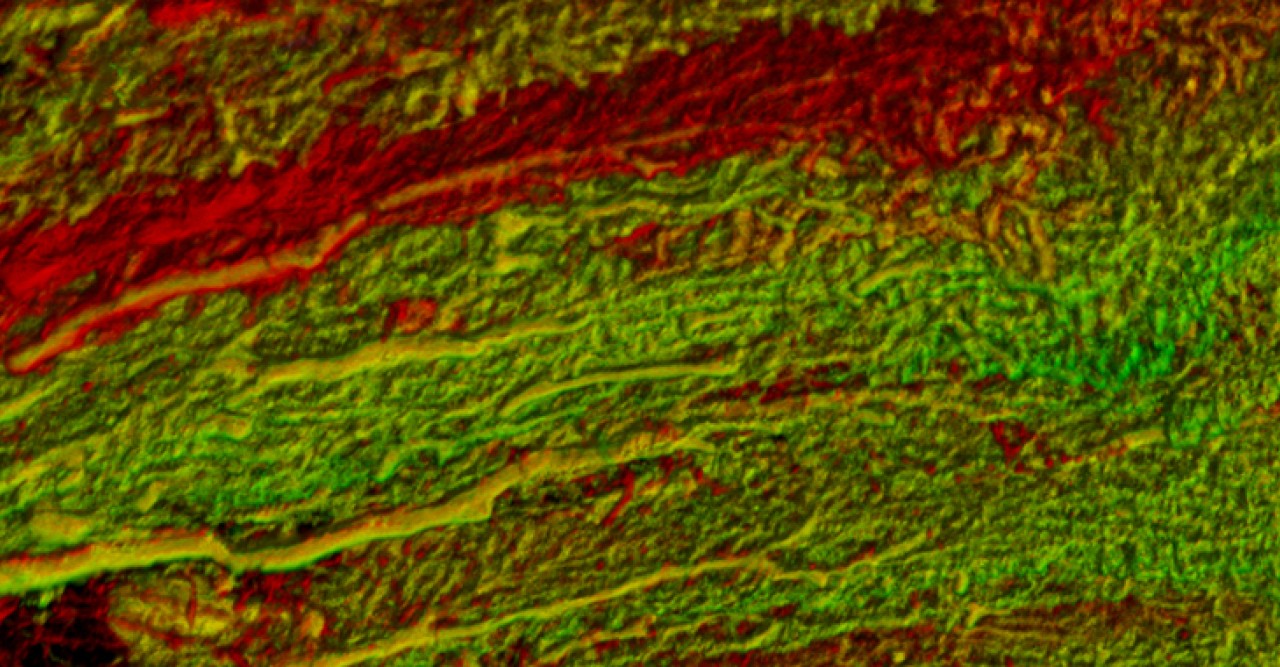
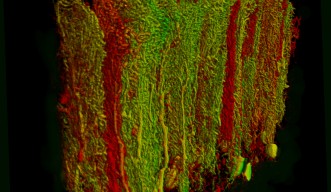
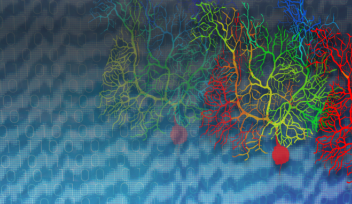
Recently, a group of researchers from Okinawa Institute of Science and Technology Graduate University and collaborators explored how changes in the activity of large neurons called Purkinje cells found in the cerebellum – the brain region known for its role in sensorimotor coordination – were correlated with these saccadic eye movements. They have recently published their results in eLife.
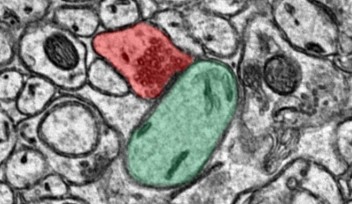
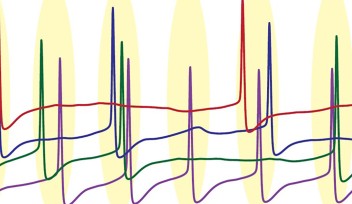
In order to do this, the team looked at the data from the brains of three live monkeys in Germany that were monitored during saccadic movement. Specifically, they were interested in how neurons transmit information, which is seen in the form of electrical signals. Just like all other neurons, Purkinje cells emit spikes caused by electrical output. Purkinje cells fire spikes rapidly most of time, but occasionally there are pauses in the spikes.
“One of the many challenges in studying neurons is to figure out the underlying cause of the different electrical signals,” Prof. Erik De Schutter, co-author and head of OIST’s Computational Neuroscience Unit said. “We want to know what these spikes are actually telling us.”
Their approach to data analysis was unique because the researchers examined not only the information regarding the spikes and pauses in the Purkinje cells, but also looked into the activity in the local field potential.
“The local field potential or the neighbourhood around a specific Purkinje cell reflects the activity in many neurons at the same time,” De Schutter said. This way, they could look at the relationship between what was happening in specific neurons and what was happening in the average activity around those neurons.
“We found that the spikes right before and after a pause showed a much larger correlation with the local field potential,” said Sungho Hong, first author and group leader of OIST’s Computational Neuroscience Unit. “That means that these spikes and pauses are not random. They are related to events happening within the larger network.”
The next step was looking at the relationship between these electrical signals in the neurons and the eye motion itself.
“There is a big change in the local field potential at the time of a saccade,” De Schutter said. “We can also see that there is a pause-beginning spike in the Purkinje cell at the time that the eye movement starts.”
The rest of the spikes that were not directly before or after a pause did not seem to have much individual importance. However, when the team built a mathematical model on the average firing rate of these spikes, they found that a simple relationship could predict the smooth aspect of motion – the part of the motion that follows after the very start of the saccade.
“This showed us that the spikes that begin the pauses control the start of a movement and that the ones that are not related to the pauses control the velocity of the movement,” De Schutter said. “This means that there is multiplexing in the Purkinje cells – they can send out two signals at once.”
This is important because this research suggests that both timings of individual spikes and the average firing rate of the spikes are crucial in understanding the complexities of the cerebellum and fine motor control.
This insight into understanding the mechanism of the neurons in the cerebellum could be useful in current medical technology, such as, brain machine interfacing, which allows paralyzed patients to move by re-routing brain signals. Additionally, this information could be useful in robotics that require fine motor control.
“Details matter,” De Schutter said. “It is easy to just look at the average, but the individual spikes reveal interesting information.”
Specialties
Research Unit
Submit a press inquiry