29 October 2015
Receptors: It Takes a Dimer to Bind
“This ‘rotation model’, which made the cover of BioEssays, represents a true paradigm shift in the membrane receptor field,” stated Prof. Pierre De Meyts, a renowned researcher of insulin and receptor binding for almost half a century and one of the reviewers of the paper by Prof. Ichiro Maruyama, the head of the Information Processing Biology Unit at Okinawa Institute of Science and Technology Graduate University (OIST).
Based on meta-analysis of over 100 studies of transmembrane cell-surface receptors, as well as his own research, Prof. Maruyama proposed a new model for receptor activation — the “rotation model”. If the model proves correct, it will lead to major changes in molecular and cell biology, biochemistry, and the pharmaceutical industry.
Cell Communication with the Outside World
Cell contents are separated and protected from the external environment by cell membranes. Survival and reproduction of living organisms depend upon proper perception and processing of environmental signals. Cell-surface receptors are proteins embedded in the cell membrane. They are responsible for communication between the cell and everything around it.
These receptors are highly specialised and respond only to specific molecules, ignoring all others. Therefore, there are many different kinds of receptors. For example, humans have over a thousand encoded in their genome. However, the basic mechanism of activation is the same for all receptors: a ligand — for example a hormone, growth factor, cytokine, or nutrient — binds to the receptor and triggers changes in the cell's metabolism and activity.
Ligand binding is a very basic biological process crucial to all functions of a living organism. Abnormal activation of receptors is often implicated in cancers, and developmental and mental diseases. Therefore, understanding of ligand binding is essential for pharmaceutical research and may result in better drugs — lower doses, greater effectiveness, and fewer side-effects.
Drugs are essentially molecules that can interact with receptors and participate in ligand binding, triggering desired cell responses. Drugs often mimic natural ligands and can be seen as part-time workers substituting for staff members who did not show up to work. Who wants workers who do not do what they were hired to do?
The Old "Dimerization Model"
According to the previous “dimerization" model, prior to ligand binding receptors exist in monomeric form. Contact with a ligand prompts receptors to bind together to form a dimer — a functional, compound receptor comprising two structurally similar monomers.
Prof. Maruyama disagrees: “If receptors existed in monomeric form it would be harmful to the cell”. Membranes of animal cells are not solid and at normal body temperature resemble vegetable oil in consistency. Proteins, cholesterol, and other substances making up the membrane’s mosaic are free to move across it. Random collisions of monomeric receptors could activate them in the absence of a ligand. "But nature is smart," he says, "therefore, the receptors must be dimers prior to ligand binding." And, indeed, previous research has shown that receptor tyrosine kinases, e.g. insulin receptors, and cytokine receptors, e.g. growth hormone receptors, exist in dimeric form even in the absence of ligands.
The "Rotation Model"
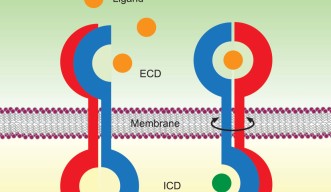
Prof. Maruyama’s “rotation model” provides an explanation of the ligand activation mechanism of dimeric receptors. He examines several well-studied receptors — the bacterial aspartate receptor (Tar), human epidermal growth factor receptor (EGFR), and human brain-derived neurotrophin receptor (BDNFR) — and comes to a conclusion that they all have similar chemical structures with or without their corresponding ligands. Their transmembrane regions, which span the cell membrane, are likely to rotate on their long axes, perpendicular to the membrane.
Ligand binding to the extracellular part of the receptor induces rotation of the receptor’s transmembrane region inside the cell membrane, and in doing so, regulates its activity inside the cell. The flexibility of the protein also changes in the process. Prior to ligand binding, the external end of the receptor is flexible, while the inside part is less flexible. After ligand binding, the extracellular portion loses flexibility while the intracellular portion gains it. The flexibility is driven by thermal energy provided by the organism’s body temperature.
Prof. Maruyama suggests that rotation of a receptor’s transmembrane region requires less energy than lateral movement of monomeric receptors inside the fluid membrane proposed by the “dimerization theory”. Considering that nature tends to choose energy-efficient designs, this is one more persuasive argument in favour of Prof. Maruyama’s model.
Prof. De Meyts assures that “Prof. Maruyama makes a convincing case that this [dimerization] model is incorrect, and that in fact, numerous cell membrane receptors are pre-dimerized in the membrane and are activated allosterically by ligand binding.”
Specialties
Submit a press inquiry