Solar cells, which convert sunlight to electricity, have long been part of the global vision for renewable energy. Although individual cells are very small, when upscaled to modules, they can be used to charge batteries and power lights. If laid side-by-side, they could, one day, be the primary energy source for buildings. But the solar cells currently on the market utilize silicon, which makes them expensive to fabricate when compared to more traditional power sources.
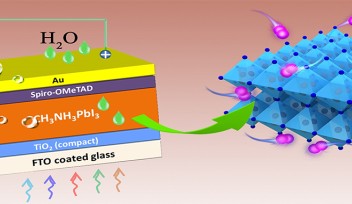
That’s where another, relatively new-to-science, material comes in – metal halide perovskite. When nestled at the center of a solar cell, this crystalline structure also converts light to electricity, but at a much lower cost than silicon. Furthermore, perovskite-based solar cells can be fabricated using both rigid and limber substrates so, alongside being cheaper, they could be more light-weight and flexible. But, to have real-world potential, these prototypes need to increase in size, efficiency, and lifespan.
Now, in a new study, published in Nano Energy, researchers within the Energy Materials and Surface Sciences Unit, led by Professor Yabing Qi, at the Okinawa Institute of Science and Technology Graduate University (OIST) have demonstrated that creating one of the raw materials necessary for perovskites in a different way could be key to the success of these cells.
“There’s a necessary crystalline powder in perovskites called FAPbI3, which forms the perovskite’s absorber layer,” explained one of the lead authors, Dr. Guoqing Tong, Postdoctoral Scholar in the Unit. “Previously, this layer was fabricated by combining two materials – PbI2 and FAI. The reaction that takes place produces FAPbI3. But this method is far from perfect. There are often leftovers of one or both of the original materials, which can impede the efficiency of the solar cell.”
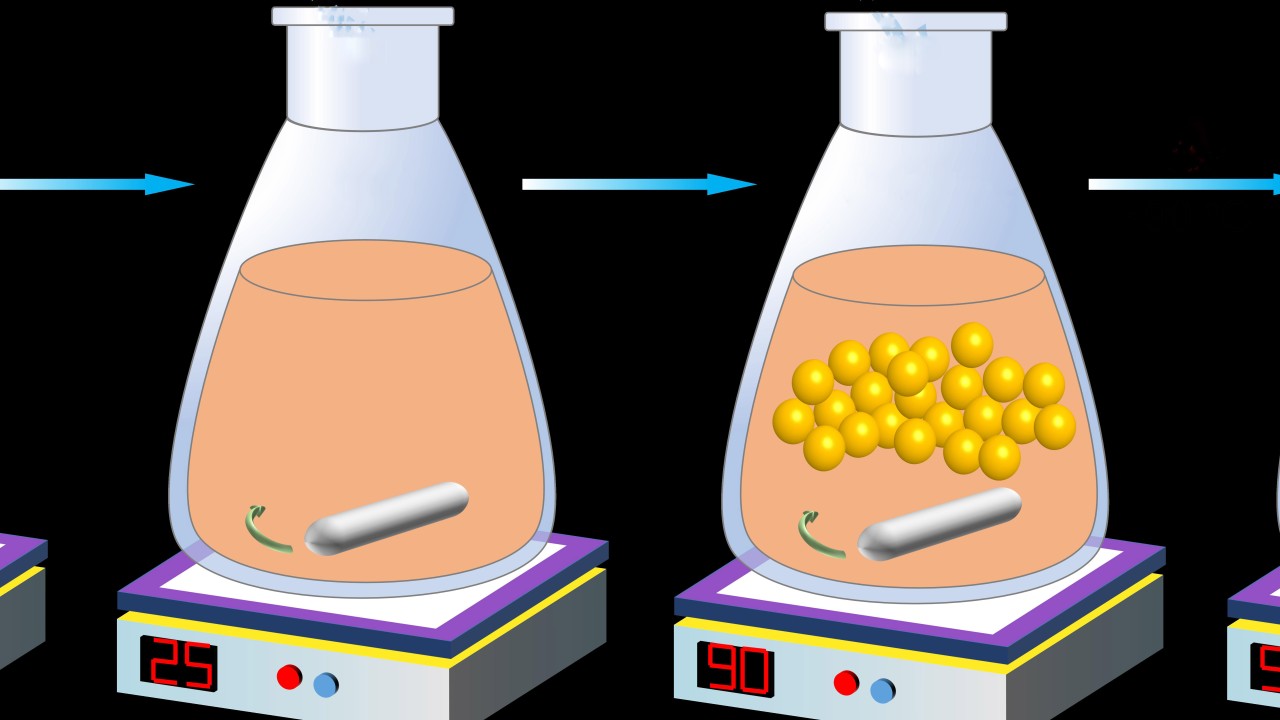
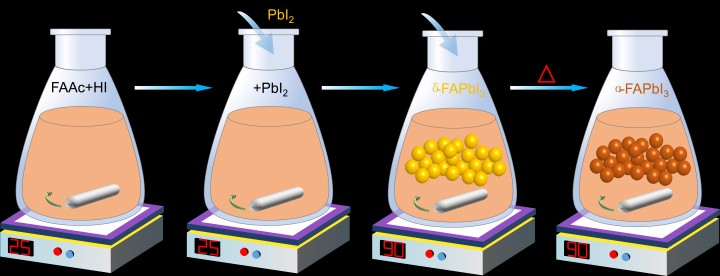
To get around this, the researchers synthesized the crystalline powder using a more precise powder engineering method. They still used one of the raw materials–PbI2— but also included extra steps, which involved, amongst other things, heating the mixture to 90 degrees Celsius and carefully dissolving and filtering out any leftovers. This ensured that the resulting powder was high quality and structurally perfect.
The researchers used a powder engineering method to create a high-quality version of FAPbI3. First, they mixed formamidinium acetate (FAAc) with hydroiodic acid (HI). PbI2 was then added. The mixture was then heated to 90 degrees Celsius. In the final step, any remaining impurities or unreacted materials were dissolved in water and filtered out.
Another benefit of this method was that the perovskite’s stability increased across different temperatures. When the perovskite’s absorber layer was formed from the original reaction, it was stable at high temperatures. However, at room temperature, it turned from brown to yellow, which wasn’t ideal for absorbing light. The synthesized version was brown even at room temperature.
In the past, researchers have created a perovskite-based solar cell with more than 25% efficiency, which is comparable to silicon-based solar cells. But, to move these new solar cells beyond the lab, an upscale in size and long-term stability is necessary.
“Lab-scale solar cells are tiny,” said Prof. Qi. “The size of each cell is only about 0.1 cm2. Most researchers focus on these because they’re easier to create. But, in terms of applications, we need solar modules, which are much larger. The lifespan of the solar cells is also something we need to be mindful of. Although 25% efficiency has previously been achieved, the lifespan was, at most, a few thousand hours. After this, the cell’s efficiency started to decline.”
The OIST Energy Materials and Surface Sciences Unit work with perovskite solar cells and modules of varying sizes.
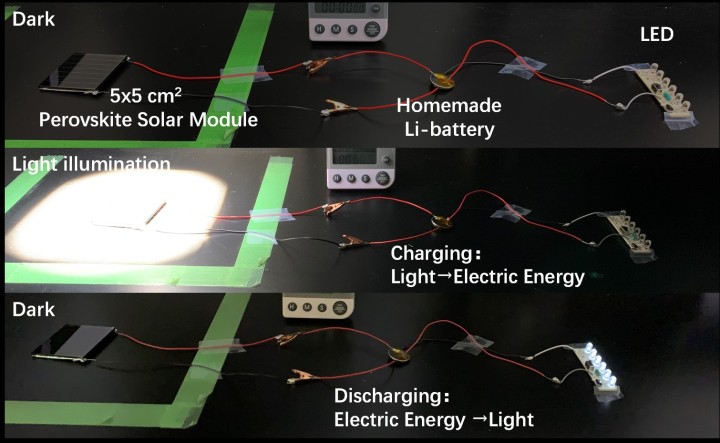
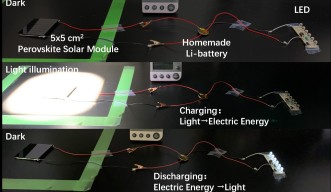
Using the synthesized crystalline perovskite powder, Dr. Tong, alongside Postdoctoral Fellow Dr. Dae-Yong Son and the other scientists in Prof. Qi’s Unit, achieved a conversion efficiency of over 23% in their solar cell, but the lifespan was more than 2000 hours. When they scaled up to solar modules of 5x5cm2, they still achieved over 14% efficiency. As a proof-of-concept, they fabricated a device that used a perovskite solar module to charge a lithium ion battery.
A proof-of-concept device uses a 5×5cm2 perovskite solar module to charge a lithium ion battery.
A proof-of-concept device created by the OIST Energy Materials and Surface Sciences Unit uses a perovskite solar module to charge a lithium ion battery.
These results represent a crucial step towards efficient and stable perovskite-based solar cells and modules that could, one day, be used outside of the lab. “Our next step is to make a solar module that is 15x15cm2 and has an efficiency of more than 15%,” said Dr. Tong. “One day I hope we can power a building at OIST with our solar modules.”
This work was supported by the OIST Technology Development and Innovation Center’s Proof-of-Concept Program.
Research details
- Journal: Nano Energy
- Paper: Removal of residual compositions by powder engineering for high efficiency formamidinium-based perovskite solar cells with operation lifetime over 2000 h
- Authors: Guoqing Tong, Dae-Yong Son, Luis K. Ono, Hyung-Been Kang, Sisi He, Longbin Qiu, Hui Zhang, Yuqiang Liu, Jeremy Hieulle, Yabing Qi
- DOI: 10.1016/j.nanoen.2021.106152