20 December 2017
Shining a Light on Bacterial Cell Division
Imagine trying to defeat an army of invaders that can double its population size every twenty minutes. This is what the human body is facing when it becomes infected with a harmful strain of Escherichia coli (E. coli), a type of bacteria which can multiply rapidly and cause a host of unpleasant and potentially dangerous illnesses, such as diarrhea, respiratory illness and pneumonia.
With the world-wide rise of antibiotic resistance, scientists are desperately seeking new ways to fight bacterial infections with drugs. One effective method of preventing bacterial cells from dividing and multiplying would be to target the cell division machinery. However, to achieve this, a more detailed picture of the structure and organization of the machinery itself is required.
Researchers from the Structural Cellular Biology Unit at the Okinawa Institute of Science and Technology Graduate University (OIST), in collaboration with researchers from Stockholm University, have shed light on the mechanism of cell division in E. coli. Their research was recently published in Molecular Microbiology.
In the long run, this research could help to identify new ways to target bacteria with antibiotics. “If we can understand the mechanisms by which bacterial cells divide in better detail, then we can try to create drugs that disrupt these mechanisms,” says Bill Söderström, lead author of the paper.
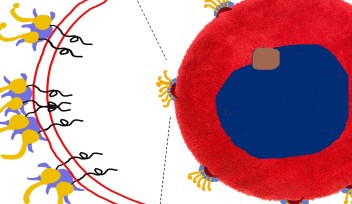
Most bacterial cells replicate by binary fission, a process in which the mother cell constricts and separates into two identical daughter cells. During cell division, a large molecular machine called the ‘divisome’ assembles inside the cell. The researchers have revealed the spatial organization of two key proteins from the E. coli divisome, ‘FtsZ’ and ‘FtsN’.
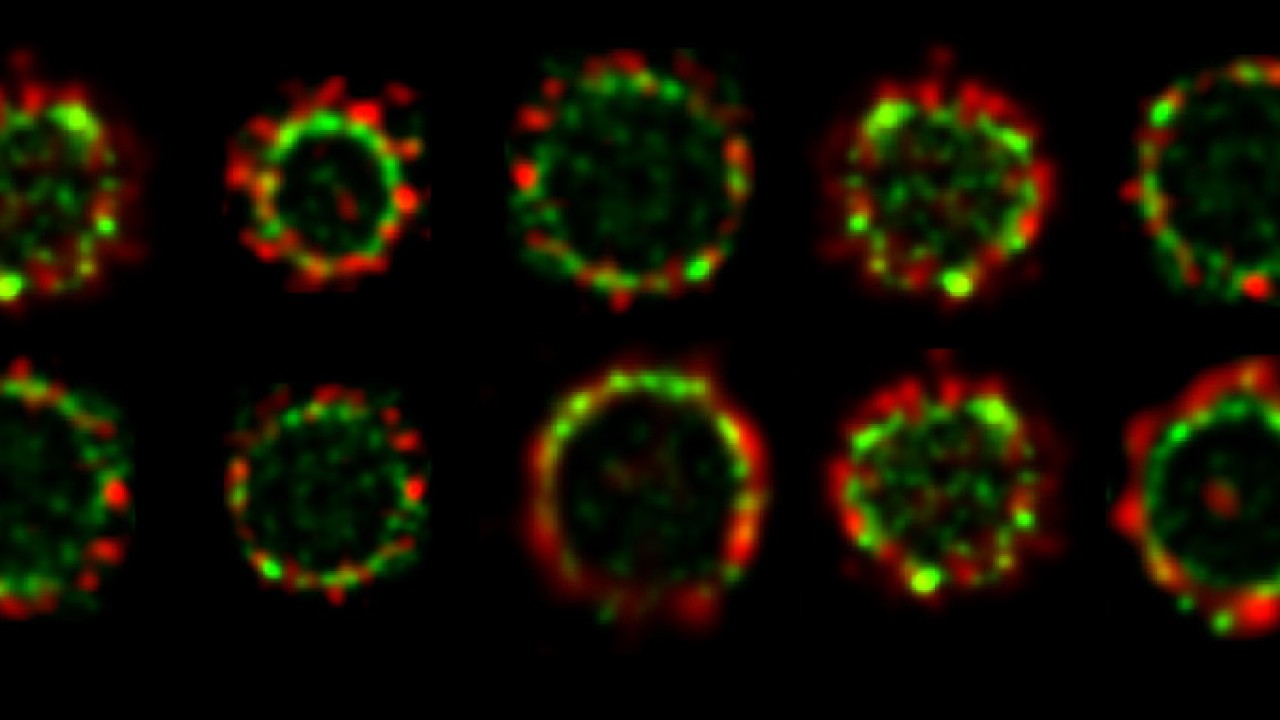
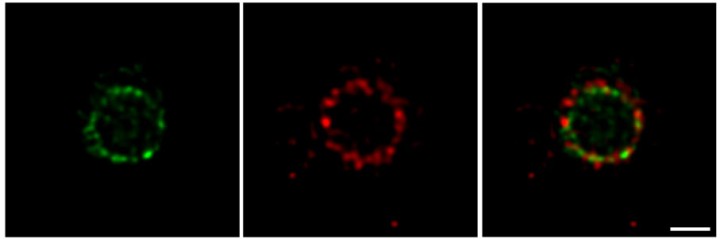
For a long time, cell biologists had assumed that all proteins in the divisome were clustered together in one large super-complex. Conventional fluorescence microscopy has relatively low resolving power, meaning that adjacent objects that are very close together sometimes appear as a single entity. However, using a cutting-edge imaging technique available at OIST called super-resolution Stimulated Emission Depletion (STED) nanoscopy, the researchers were able to visualize the division machinery at the nanoscale. “With better resolution, we were able to see the difference between the two protein rings and to infer details about the process of cell division,” says Söderström.
Using two florescent colors to label FtsZ and FtsN in green and red respectively, the researchers revealed that both proteins are localized in large assemblies, which are distributed unevenly around the division site. Early in the division process, the two proteins form non-overlapping patchy rings. As cell division progresses, the green ring, formed by FtsZ, moves inside the red ring, formed by FtsN. The finding that these proteins do not always overlap but are separated into several groups, suggests that the divisome does not operate as a single molecular machine. Rather, each protein group plays a specific role.
With a more detailed picture of the cell division machinery, biologists can design new antibiotics to prevent bacterial cells from dividing and multiplying. “The next step is to look at many more pairs of cell division proteins and to figure out which of these we should target with drugs,” says Söderström.
Research Unit
Submit a press inquiry