27 January 2020
Synchronized Swimming: Biology On a Micro-Scale
Specialized stringy fluids flow through the human joints and help constitute substances such as mucus. These fluids contain long, flexible molecules like polymers or proteins, giving them the ability to stretch and absorb shock.
However, scientists have yet to fully understand how these enigmatic fluids interact with small-scale biological structures. Structures of particular interest are cilia – tiny hair-like projections attached to the cell membrane, which undulate to perform functions such as clearing contaminants out of airways. These fluid-structure interactions are important for understanding precisely how cilia move to perform their biological duties. However, these interactions occur on such a small scale that they have been difficult to study experimentally.
Now, researchers in the Micro/Bio/Nanofluidics Unit at the Okinawa Institute of Science and Technology Graduate University (OIST) have identified some key features of how these so-called viscoelastic fluids flow around cilia. Viscoelastic fluids are viscous, like molasses, as well as stretchy. The study, published in Small, suggests that it is the fluids’ elasticity that drives the patterned movement of the cilia, the researchers say.
Entering the world of the very small
To set up their experiment, the scientists hollowed out microchannels in fused silica glass. These channels contained either one or two flexible cylindrical posts attached to one side of the channel, which represented cilia.
The scientists then used syringe pumps to propel a viscoelastic solution through the glass microchannels at a precisely controlled speed. The experimental fluid contained wormlike micelles (also called living polymers), which are micron-sized flexible structures that mimic the movement of biological molecules present in human body fluids.
The researchers took a series of measurements, using three separate high-powered microscopes with different optical techniques to capture the behavior and properties of the fluid as it interacted with the posts.
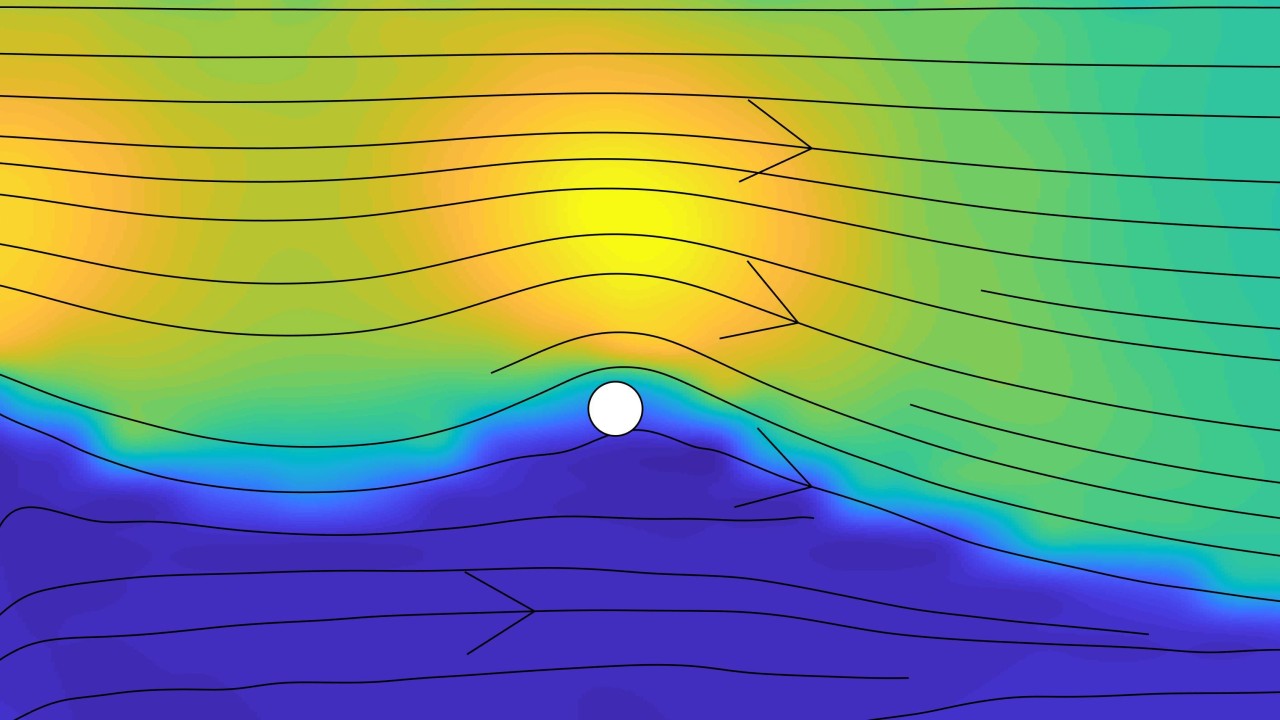
First, the scientists used a method called micro-particle image velocimetry to record the velocity of the fluid as it flowed around the posts. They observed that the fluid preferentially moved around one side of the posts, leaving virtually stationary fluid on the other side. At certain flow speeds, however, the fluid on the stationary side began flowing in a jerky motion.
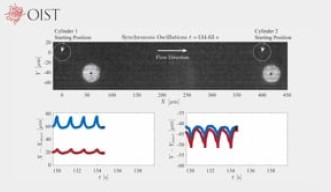
As fluid moved, the post began oscillating. “An important aspect of the study was our ability to carefully track the resulting oscillations of the posts as a function of time using high-speed video microscopy”, said Dr. Simon Haward, the group leader of the unit.
Using a method called high-speed polarized light microscopy they were also able to trace the regions around the cylindrical posts where the worm-like micelles stretched elastically, and to correlate the amount of stretching with the position of the posts.
While interacting with the fluid, two posts located near one another began to oscillate in almost perfect synchrony, suggesting that the fluid elasticity is mediating the synchronous beating of a cell’s cilia, the researchers say.
“The synchronous time-dynamics of the posts are completely imparted by the fluid itself,” said Dr Cameron Hopkins, the first author of the study. “However, this only happens under specific conditions. If we increase the flow rate and thus the influence of the fluid’s elasticity, then we lose the regularity of the oscillations and it becomes erratic.”
Developing new biological models
Moving forward, the scientists hope to study how changing the flexibility and distances between the cylindrical posts will affect their behavior. Hopkins and his colleagues also hope to repeat the experiment in a larger system with up to twenty cylindrical posts to emulate an array of cilia.
“Our current experimental setup is an idealized geometry -- of course, real biological systems are much more complicated,” said Professor Amy Shen, the head of the Micro/Bio/Nanofluidics Unit. “This current model is a stepping-stone to something more complex and more biologically relevant.”
The researchers hope that further research will help illuminate the physics of the very small – and perhaps provide insight into the dynamic movements occurring within our very cells.
Specialties
Research Unit
Submit a press inquiry