12 May 2014
Studying Behavior Using Light to Control Neurons
A new paper published by OIST’s Neurobiology Research Unit identifies some of the neurons responsible for behavioral decisions in rats. Using a technique that employs light to control nerve cell activity, researchers in Professor Jeff Wickens’ Unit inactivated a region of the brain and showed that it caused the rats to behave more flexibly while trying to get a reward. The technique, called optogenetics, allows researchers to “show that the firing or inhibition of certain neurons has a causative relationship with a given behavior, whereas previous methods only allowed us to correlate neuronal activity with behavior,” says Prof. Wickens. The paper, recently published in Learning & Memory, describes the group’s work using optogenetics to study behavioral flexibility.
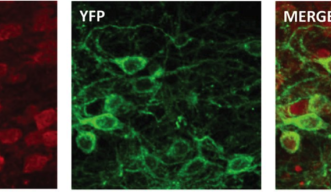
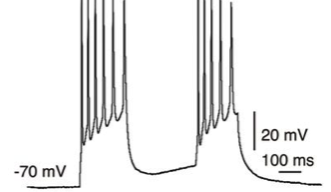
Optogenetics works by causing certain neurons in the brain to express a protein that is sensitive to light. The researchers then shine a light into the brain using a fiber optic cable and can control the activity of the neurons expressing the light-sensitive protein. In the paper, researchers described how they used this method to prevent a set of neurons from firing, or inhibiting their activity. This method allows specific targeting within the brain and tight control of the timing of activating or inactivating neurons. Optogenetics is “turning neuroscience upside down,” according to Wickens.
In the publication, the researchers describe the influence of an area of the brain called the nucleus accumbens and its role in behavioral flexibility, or the ability to change strategies during a task. Rats were given a choice between pushing one of two levers. They were rewarded when they pushed one lever, and continued to receive a reward pushing the lever up to 20 times. On the 21st time, the lever stopped delivering a reward, and the other lever was then programmed to deliver the reward. Normally, rats continue to press the unrewarded lever several times before switching strategies and pushing the other lever to receive a reward. However, when the nucleus accumbens shell neurons were turned off, using optogenetic inhibition, the rats changed their behavior more quickly and began pushing the other lever sooner. Inhibition of the neurons only caused this behavioral shift when the neurons were turned off at the time of feedback of results, when the rats found out if they pressed the rewarded lever. In other words, the rats did better, made the switch to the other lever sooner, when these neurons were silenced at a specific time.
The work is the first to show that optogenetic inhibition of the nucleus accumbens shell neurons during reward or error feedback increases behavioral flexibility. Wickens says the work “suggests that neurons integrate a history of reward and animals consult those neurons on whether to continue or change strategies.” It appears the nucleus accumbens shell neurons are some of the neurons that hold this history of rewards. Showing the clear cause and effect of the inhibition of these neurons with this decision-making behavior would not have been possible without optogenetics.
Looking forward, Wickens is excited about the possibilities that optogenetics holds. “The real challenge of understanding how the brain works is that it’s been hard to identify the neural activity causing specific behaviors. With this technique, we can now do that.”
You can read the paper in Learning & Memory here:
http://learnmem.cshlp.org/content/21/4/223.long
By: Kathleen Estes
Specialty
Research Unit
Submit a press inquiry