Brain Mechanisms for Behaviour Unit
Principal Investigator: Gordon Arbuthnott
Research Theme: A study of the brain regions primarily affected in Parkinson’s disease as a way to understand initiation of spontaneous behavior.
Abstract
1. Staff
- Dr. Marianela Garcia Munoz, Group Leader
- Dr. Fiona Randall, Researcher (2008-2010)
- Dr. Takuya Hikima, Researcher
- Dr. Luis Alberto Carrillo-Reid, Researcher (2009-2011)
- Omar Pedro Jaidar Benavides, Short-term research assistant
- Chidinma Chikaodinaka Nwigwe, Short-term research assistant
- Ms. Hiroko Chinone, Research Administrator/Secretary
2. Collaborations
- Theme: In vitro isolation of genetically marked neurons.
- Type of collaboration: Joint research
- Researchers:
- Professor W.A. Staines, University of Ottawa
- Professor A. Krantis, University of Ottawa
Centre for Research in Biophrmaceuticals & Biotechnology (CRBB) - Professor Geoff Mealling, Senior Research Officer, Institute of Biological Sciences
National Research Council of Canada - Dr. Sarah Schock, University of Ottawa
- Kheira Jolin-Dahel, M. Sc., University of Ottawa
3. Activities and Findings
3.1 Professional Skills for Scientists
A course on “Professional Skills for Scientists” was organized by the Unit. Prof Michael Zigmond and Dr Beth Fisher facilitated a three-day seminar series guiding the group through relevant topics/skills required in the development of a scientist; grant writing, presentation of results, awareness of ethics and preparation and publication of manuscripts. Also very welcomed was the topic on how to be successful on job applications and job interviews. In general, the course was well received by the staff and students who attended. Course evaluations were very good. Moreover the course highlighted a basic need for future OIST graduate training. Prof Zigmond also presented his research results on endogenous neuroprotection mechanisms in an animal model of Parkinson’s disease.
Figure 1: Participants in the Professional Skills course.
Professor Zigmond and Dr Fisher are in the centre of the front row.
3.2 IBAGS X
The International Basal Ganglia Society meeting takes place every three years. IBAGS X took place in New York State. Unit’s posters were up for the duration of the meeting allowing plenty time for discussion of our results and methods with other experts in the area.
3.3 Properties of corticostriatal pairs of neurons
The previous output of IBAGS meetings has been a book. For IBAGSX, however, it was decided to use the Frontiers Journal Series to publish a ‘Special Topic’ within the Frontiers in Neuroscience. The new format has the advantage of having the manuscripts critically reviewed rather than just edited by book editors. The paper from the Unit has been accepted for publication. It contains the first description of the properties of corticostriatal pairs of neurons in mixed cultures. As mentioned in our last report, this is not an ideal culture because of anomalous striatocortical synaptic activity. However, the cortical neurons do make the expected excitatory synapses on their striatal neighbors and those results are the focus of our report. Nonetheless we also illustrate the anomalous connections that are only seen in mixed cultures.
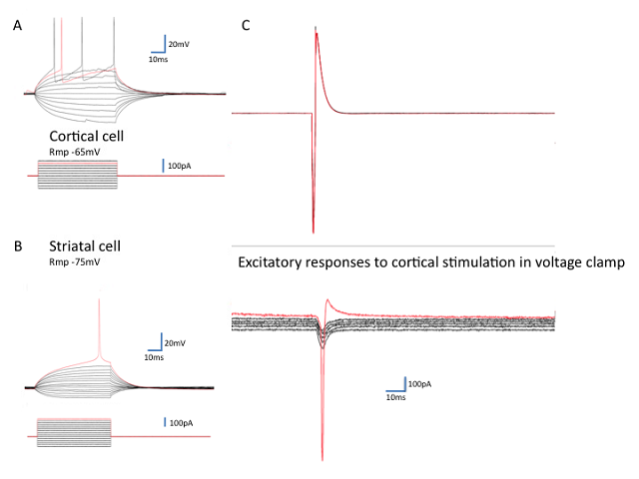
Figure 2: Corticostriatal responses in mixed cultures.
Mixed cultures of cortical neurons from transgenic mice expressing green fluorescent protein (GFP) with striatal cells from wild-type mice of the same developmental stage (E14.5) allowed visual identification of individual corticostriatal pairs. A and B show typical current clamp recordings of cortical and striatal neurons maintained from 12-37 days in vitro (action potentials at threshold are shown in red). On the right is a display of the excitatory postsynaptic currents (EPSCs) seen in response to the action current in the cortical neuron shown in the top red trace In voltage clamp mode the resting membrane potential of the striatal cell was decreased from -80mv to -30mV in steps of 10mV. The EPSCs remain inward until the cell fires an action current (not adequately clamped).
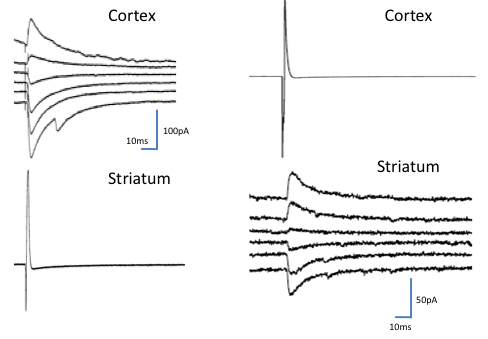
Figure 3: Synaptic responses only seen in culture.
Striatal cells induce inhibitory currents in cortical cells (A) and some cortical cells inhibit striatal ones (B). Inhibitory postsynaptic currents reverse in direction at about the chloride equilibrium potential. Voltage steps in the postsynaptic cell are as for Figure 2 (-80mV to -30mV in 10 mV steps).
3.4 Corticostriatal activity of cortical and striatal neurons grown in separate compartments
Throughout the past year we have continued to develop cultures of segregated sets of neurons from cortex and striatum. These cultures allow the development of corticostriatal connections. Patch-clamp recordings from either side of the 500µm gap in cultures is now possible. Moreover, when the cells are plated on multielectrode arrays it is also possible to record their electrical activity. We have used these segregated cortical and striatal cultures to study the pharmacology of their spontaneous activity before and after cutting the connections between them. We have also collaborated with the Wickens Unit using transfected dissociated cells to study the system with optogenetic methods. We intend to publish the method and some preliminary results while we develop the details of the experiments themselves.
In addition, we have compared the results of multielectrode array records and calcium imaging of the same cultured cortical neurons. We have a manuscript in revision that -for the first time- shows the development of compositional properties and demonstrates that neuronal groups fire in the same sequence at different time scales.
This year, we had a paper published in the Frontiers series (Garcia-Munoz et al., 2010) that has already been sited in a clinical research paper regarding the EEG changes in patients with Parkinson’s disease (Swann et al., 2011)
3.5 Presynaptic activity
Dr Hikima has established the synaptopHluorin transgenic mouse strains in OIST and we have begun to study release from individual terminals in slices from the brains of these mice. We are also developing lentiviral carriers for plasmids consisting of synaptopHluorin and a more sensitive vesicular release marker SynpHluorin in order to be able to monitor transmitter release from cultured neurons.
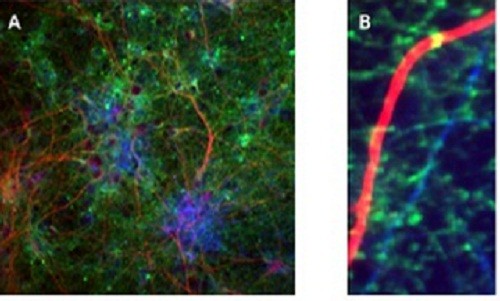
Figure 4: Antibody staining of a transfected culture of cortical neurons.
The culture was stained with anti-NF160 (to stain axons blue); anti-MAP2 (to stain dendrites red) and with a monoclonal antibody to eGFP which stains the synaptopHluorin green. (A) shows a general view of the culture while at high power (B) green vesicle-containing dots are visible along the blue axons and individual synapses are visible on the red dendrite as yellow dots.
3.6 A mouse model for Parkinson’s disease
During the year we had two very productive student visits. Omar Pedro Jaidar Benavides visited from the National Autonomous University of Mexico and Chidinma Chikaodinaka Nwigwe (Kim) came from Abia State University, in Nigeria.
While Omar was with us he developed not only the methods for creating a mouse model of parkinsonism (Figure 4), but also completed publication of a significant paper on a similar rat model with Luis Carrillo-Reid (Jaidar et al., 2010). We look forward to Omar’s return as soon as he completes his Ph.D graduation in Mexico in the next few months.
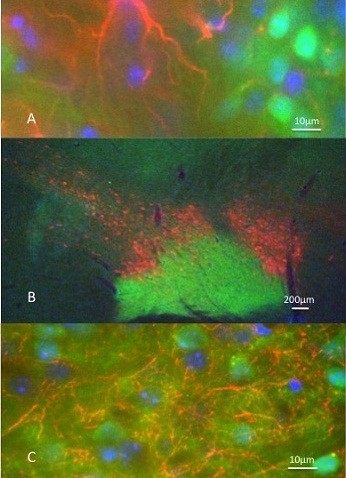
Kim was in the final year of her preclinical medical studies and opted to do some research work with us. Her visit also produced excellent histochemical work on a similar theme. Although she did not have any previous laboratory experience, she left with some interesting results included in Figure 5. Striatal dopamine depletion elicits dystrophic changes in dendrites of medium spiny neurons. Previous work from my laboratory (e.g., Ingham et al. 1989) and others have reported a marked decrease in striatal spine density following loss of striatal dopamine. Kim provided evidence that the damaged striatum has a clear astrogliosis and documented the distribution of several antigens in the normal and dopamine-lesioned brains.
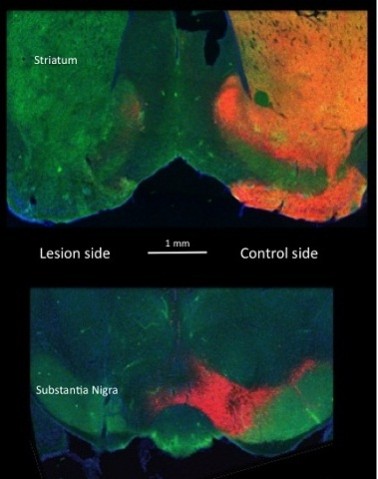
Figure 5: Evidence of the success of a unilateral lesion of substantia nigra dopamine cells in a Drd1-EGFP BAC transgenic mouse.
This figure shows brain sections containing the substantia nigra and striatum of bacteria artificial chromosome (BAC) transgenic mice expressing the reporter protein enhanced green fluorescent protein (EGFP) under the control of the D1 dopamine receptor promoter (Drd1-EGFP). Two parts of brain sections are shown. The upper photomicrograph from the forebrain striatal area shows on the right bright orange colour reflecting the presence of coexisting tyrosine hydroxylase (TH, stained red) and GFP (stained green). The absence of the red on the left side indicates the loss of striatal dopamine and displays only the Drd1 staining in the striatal cells. In the lower part of the figure, the area of the substantia nigra is shown. The red cells on the right are the intact dopamine-containing neurons that give rise to the TH staining in the striatum above. On the left of the section again the absence of the red indicates the loss of dopamine-containing neurons (no TH). The area is only marked by the EGFP indicating the presence of dopamine D1 presynaptic receptors in the axon terminals of striatal projections to substantia nigra pars reticulata. The intact GFP staining on the left provides an indication that striatal neurons still make normal projections to the area in the absence of dopamine.
Figure 6: Further anatomical studies of BAC mice following a unilateral lesion substantia nigra dopamince cells.
The three pictures above represent known properties of dopamine neurons in rat brain that we confirmed in transgenic BAC mice expressing Drd1-EGFP. Namely: 1-After a lesion of dopamine-containing neurons there is proliferation of glia in the striatum on the lesioned side. A-Striatal GFAP expression in astrocytes (red) can be seen in the transgenic mouse mingled with neurons expressing the D1 dopamine receptor promoter (green) and the nucleic acid staining DAPI (4',6' diamino-2-phenylindole·2HCl) (blue). 2-Dopamine cells extend their distribution beyond the anatomical borders of substantia nigra. B- In this sagittal section from an intact Drd1 mouse the anterior posterior distribution of dopamine cells around the Drd1-labeled nigra is clearly illustrated. 3-Dopamine-containing axons widely distribute among striatal cells. C) The high power micrograph shows the individual dopamine axons (red) among the Drd1 expressing cells (green). The other striatal cells are shown with blue nuclear staining (DAPI). Note: These illustrations were obtained from animals prepared by Omar (as seen in Figure 5) but the fixation, cutting and staining were all part of Kim’s training.
3.7 Anatomy of mixed cortico-striatal cultures
Current outputs from our collaborative research with Canada include the first description of the anatomy of mixed cortico-striatal cultures with an analysis of the frequency of the occurrence of interneurons (Schock et al., 2010). We have a paper on the development of cortical cultures in revision, and another on the influence of cortical cells on the development of striatal neurons that is in a final stage of preparation for submission. This topic has suddenly become relevant with the recent publication about similar developmental effects of activity on cortical interneurons (De Marco Garcia et al., 2011).
Our first attempt to isolate GFP expressing cholinergic cells failed due to a delayed expression of the marker. We are in the process of developing ways to isolate cells in which the GFP is expressed in embryos.
References
De Marco Garcia NV, Karayannis T, Fishell G (2011) Neuronal activity is required for the development of specific cortical interneuron subtypes. Nature 472:351-355.
Garcia-Munoz M, Carrillo-Reid L, Arbuthnott GW (2010) Functional Anatomy: Dynamic states in basal ganglia circuits. Frontiers in Neuroanatomy 4.
Jaidar O, Carrillo-Reid L, Hernandez A, Drucker-Colin R, Bargas J, Hernandez-Cruz A (2010) Dynamics of the Parkinsonian Striatal Microcircuit: Entrainment into a Dominant Network State. J Neurosci 30:11326-11336.
Schock S, Jolin-Dahel K, Schock P, Staines W, Garcia-Munoz M, Arbuthnott G (2010) Striatal interneurons in dissociated cell culture. Histochemistry and Cell Biology 134:1-12.
Swann N, Poizner H, Houser M, Gould S, Greenhouse I, Cai W, Strunk J, George J, Aron AR (2011) Deep Brain Stimulation of the Subthalamic Nucleus Alters the Cortical Profile of Response Inhibition in the Beta Frequency Band: A Scalp EEG Study in Parkinson's Disease. J Neurosci 31:5721-5729.
4. Publications
4.1 Journals
- Garcia-Munoz M, Carrillo-Reid L, Arbuthnott GW. Functional Anatomy: Dynamic states in basal ganglia circuits. Frontiers in Neuroanatomy 4(2010).
- Schock SC, Jolin-Dahel KS, Schock PC, Staines WA, Garcia-Munoz M, Arbuthnott GW. Striatal interneurons in dissociated cell culture. Histochemistry and Cell Biology 134, 1-12 (2010).
4.2 Books and Other one-time publications
- Arbuthnott, G. W. & Garcia-Munoz, M. Neuropharmacology. in Companion to Psychiatric Studies 8th Edition (eds. E. Johnstone, D.C. Owens, S. M. Lawrie, A. M. McIntosh, & M. Sharpe) 45-76 (Churchill Livingstone Elsevier, Edinburgh, 2010).
- Arbuthnott, G. W. & Garcia-Munoz, M. Neuropharmacology Chapter 3. in Companion to Psychiatric Studies (Churchill Livingstone, Edinburgh, 2010).
- Wickens, J. R. & Arbuthnott, G. W. Gating of cortical input to the striatum. in Handbook of Basal Ganglia Structure and Function (eds. H. Steiner & K.Y. Tseng) (AP, 2010).
4.3 Oral and Poster Presentations
- Arbuthnott, G. Growing brain cells in a dish - how not to do neuroscience?, International Brain Research Organization School of Neuroscience, Hong Kong, Jun 1, 2010
- Gordon Arbuthnott, M. G.-M., Fiona Randall, William Staines. The corticostriatal system in cell culture - some results - some nasty lessons., Department of Neurology, University of Dusseldorf, Germany, Jun 28, 2010
- Gordon Arbuthnott Growing brains in a dish – a stupid idea – or a way to answer specific questions?, Otago University, Dunedin, New Zealand, December 10, 2010.
- Hikima, T. A., Rikita; Ishizuka, Toru; Yawo, Hiromu. Synaptic vesicle dynamics and its plasticity, The 87th Annual Meeing, The Physiological Society of Japan, Morioka, Japan, May 20, 2010
- Luis Carrillo-Reid, M. G. M., Gordon Arbuthnott. Corticostriatal dynamics., Department of Neurology, University of Dusseldorf, Germany, Jun 28, 2010
- Carrillo-Reid Luis, G.-M. M., Arbuthnott Gordon. Cell assembly organization in basal ganglia circuits., International Basal Ganglia Meeting 10, Long Branch, NJ, U.S.A., Jun 21, 2010
- Carrillo-Reid Luis, G.-M. M., Arbuthnott Gordon. Unfolding the Properties of Biological Neural Networks with Multidimensional Reduction Techniques and Graph Theory., 7th International meeting on substrate-integrated microelectrode arrays, Reutlingen, Germany, Jul 01, 2010
- Fiona Randall, G. A., William Staines, Marianela Garcia-Munoz. Electrophysiological characterisation of the corticostriatal projection in culture., International Basal Ganglia Meeting 10, Long Branch, NJ, U.S.A., Jun 21, 2010
- Garcia-Munoz Marianela, C.-R. L., Arbuthnott Gordon. Properties of in vitro coupled cortico-striatal networks., International Basal Ganglia Meeting 10, Long Branch, NJ, U.S.A., Jun 21, 2010
- Hikima, T. A., Rikita; Ishizuka, Toru; Yawo, Hiromu. Synaptic vesicle dynamics and its plasticity, Dynamic modulation of synaptic activity, The 87th Annual Meeting, The Physiological Society of Japan, Morioka, Japan, Sep 01, 2010
- Randall FE, W. M., Arbuthnott GW, Cunningham MO. GluR5-containing kainate receptors drive presistent gamma frequency oscillations in the rat basolateral amygdala in vitro., Society for Neuroscience Abstract, San Diego, CA, U.S.A., Nov 15, 2010
- Takuya Hikima, M. G.-M., William Staines, Gordon Arbuthnott. Synaptic vesicle dynamics in hippocampus and striatum., International Basal Ganglia Meeting 10, Long Branch, NJ, U.S.A., Jun 21, 2010
- William Staines, S. C. S., Kheira Jolin-Dahel, Gordon Arbuthnott, Marianela Garcia-Munoz. The influence of cortical innervation on axodendritic maturation of striatal interneurons., International Basal Ganglia Meeing 10, Long Branch, NJ, U.S.A., Jun 21, 2010
5. Intellectual Property Rights and Other Specific Achievements
No report.
6. Meetings and Events
6.1 Seminar
- Title: Self Assembled Nanostructures and Nanodevices
- Date: March 15, 2011
- Venue: D015 Meeting Room, New Campus
- Speakers: Dr Simon Brown, Department of Physics, University of Canterbury, New Zealand
- Other remarks: Simon is interested in fabrication of multielectrode arrays and has developed interesting self-assembly methods for making electrodes.
6.2 Seminar
- Title: Collateral Inhibition in Neostriatum: Effects in the Neuronal Population
- Date: February 2, 2011
- Venue: D015 Meeting Room, New Campus
- Speaker: Violeta Gisselle Lopez-Huerta Ph.D. student, Biomedical Research (Neuroscience), Institute for Cellular Physiology, National Autonomous University of Mexico, Mexico City.
- Other remarks: Violeta was recommended to us by her supervisors as a possible postdoctoral researcher. Her presentation at OIST was very impressive. She will be hired upon her graduation in July, 2011.
6.3 Seminar
- Title: A Novel Neuro-Tracing Method to Visualize Presynaptic Neurons Connecting to a Single Postsynaptic Neuron
- Date: October 15, 2010
- Venue: C016 Meeting Room, New Campus
- Speaker: Takuma Mori, Ph.D., Assistant Professor, Department of Information Physiology, National Institute for Physiological Sciences, Okazaki, Aichi
- Other remarks: We intend this to be the beginning of a fruitful collaboration.
6.4 Seminar
- Title: Triggering Endogenous Neuroprotective Processes Through Exercise in Models of Dopamine Deficiency
- Date: May 11, 2010
- Venue: B250 Lecture Hall, New Campus
- Speaker: Professor Michael Zigmond, University of Pittsburgh, U.S.A.
- Other remarks: A report on Professor Zigmond's personal research.
6.5 Professional Skills for Scientists
- Date: May 12 - 14, 2010
- Venue: B250 Lecture Hall, New Campus
- Co-organizer: HR Operations section
- Speakers: Professor Michael Zigmond, University of Pittsburgh, U.S.A.
Dr Beth Fischer, University of Pittsburgh, U.S.A. - Other remarks: Dr Ichiro Maruyama assisted with the course.
7. Others
7.1 Schools outreach
- Dr. Arbuthnott talked to Yamada Junior High School students on December 10, 2010.
- Dr Yamada, Dr Doya and Dr. Arbuthnott took part in the discussion after the Science film show of “Awakenings” on March 29, 2011.
7.2 Student involvement
During the student week 4S, Dr. Arbuthnott presented on Unit’s research work. In addition a facilitated discussion was had in relation to outstanding questions in neuroscience.
7.3 Administrative support
Dr. Arbuthnott was PI representative during the development of the accounting section of OIST’s enterprise resource planning (ERP) selection committee.
7.4 Staff changes
Dr Randall left to join GlaxoSmithKline in Shanghai and Dr Carrillo-Reid left for Northwestern University in Chicago. Violeta Giselle Lopez Huerta and Omar Pedro Jaidar Benavides will join the Unit upon their Ph.D. graduation in Biomedical Research (Neuroscience) from the Institute for Cellular Physiology, National Autonomous University of Mexico, Mexico City.