Optical Neuroimaging Unit
Assistant Professor Bernd Kuhn
Abstract
In the FY 2014 the Optical Neuroimaging Unit continued and completed several imaging projects. Most research projects focused on imaging neuronal activity in the mouse brain.
1. Staff
- Dr. Bernd Kuhn, Assistant Professor
- Dr. Akihiro Funamizu, Postdoc (shared with Doya Unit)
- Dr. Christopher J. Roome, Postdoc
- Dr. Sigita Augustinaite, Postdoc
- Dr. Shinobu Nomura, Postdoc
- Ray X. Lee, Ph.D. student
- Neil Dalphin, Ph.D. student
- Leonidas Georgiou, Ph.D. student
- Dr. Kazuo Mori, Technical Assistant
- Hiroko Chinone, Research Administrator
2. Collaborations
- Theme: Model-based decision making by two-photon microscopy
- Type of collaboration: Joint research
- Researchers:
- Professor Dr. Kenji Doya, Neural Computation Unit, OIST
- Dr. Akihiro Funamizu, Neural Computation Unit and Optical Neuroimaging Unit, OIST
- Theme: NMDA spikes in dendrites of thalamocortical neurons
- Type of collaboration: Joint research
- Researchers:
- Professor Dr. Paul Heggelund, University of Oslo, Norway
- Dr. P. Johannes Helm, University of Oslo, Norway
- Theme: Multi-photon excited luminescence of magnetic FePt core-shell nanoparticles
- Type of collaboration: Joint research
- Researcher:
- Dr. Klaus Seemann, Technical University Munich, Germany
- Theme: Two-photon imaging of ascidians
- Type of collaboration: Joint research
- Researcher:
- Professor Dr.Takeo Horie, Shimoda Marine Research Center, University of Tsukuba, Japan
- Theme: Dendritic diameters affect the spatial variability of intracellular calcium dynamics in computer models
- Type of collaboration: Joint research
- Researcher:
- Dr. Haroon Anwar, Computational Neuroscience Unit, OIST
- Dr. Hermina Nedelescu, Computational Neuroscience Unit, OIST
- Dr. Weiliang Chen, Computational Neuroscience Unit, OIST
- Prof. Dr. Erik De Schutter, Computational Neuroscience Unit, OIST
3. Activities and Findings
3.1 Chronic Cranial Window with Access Port
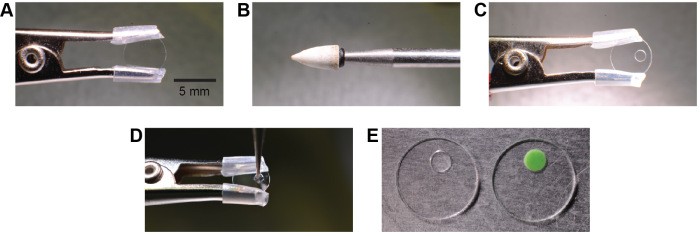
Chronic cranial windows have been instrumental in advancing optical studies in vivo, permitting long-term, high-resolution imaging in various brain regions. However, once a window is attached it is difficult to regain access to the brain under the window for cellular manipulations. Here we describe a device that combines long term in vivo optical imaging with direct brain access via glass or quartz pipettes and metal, glass, or quartz electrodes for cellular manipulations like dye or drug injections and electrophysiological stimulations or recordings while keeping the craniotomy sterile. Our device comprises a regular cranial window glass coverslip with a drilled access hole later sealed with biocompatible silicone. This chronic cranial window with access port is cheap, easy to manufacture, can be mounted just as the regular chronic cranial window, and is self-sealing after retraction of the pipette or electrode. We demonstrate that multiple injections can be performed through the silicone port by repetitively bolus loading calcium sensitive dye into mouse barrel cortex and recording spontaneous cellular activity over a period of weeks. As an example to the extent of its utility for electrophysiological recording, we describe how simple removal of the silicone seal can permit patch pipette access for whole-cell patch clamp recordings in vivo. During these chronic experiments we do not observe any infections under the window or impairment of animal health.
This technique was published in Frontiers in Cellular Neuroscience. PCT patent application was submitted in FY2013.
3.2 Action-Dependent State Prediction in Mouse Posterior Parietal Cortex during an Auditory Virtual Navigation Task
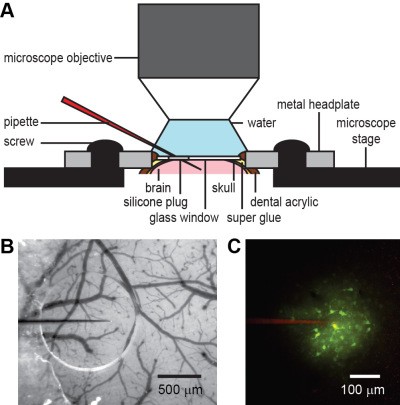
Model-based decision making requires representation of predicted states that are updated by action-dependent state transition models. To investigate their neural implementation, mice were trained to do an auditory virtual navigation task and neuronal activity was recorded in the posterior parietal cortex (PPC) and its sensory upstream, the secondary visual cortex (V2), with the genetically encoded calcium indicator GCaMP6f after gene transfer by AAV2/1 and 2-photon microscopy (Fig. A).
A mouse was head restrained and maneuvered a spherical treadmill. 12 speakers around the treadmill provided an auditory virtual environment. The direction and amplitude of sound pulses emulated the location of the sound source, which was moved according to the mouse’s locomotion on the treadmill. When the mouse reached the sound source and licked a spout, it got a water reward. The task consisted of two conditions: continuous condition in which the guiding sound was presented continuously and intermittent condition in which the sound was presented intermittently.
In both conditions, mice increased lickings as they approached the sound source, indicating that mice recognized the sound-source position and predicted a reward. In intermittent condition, the anticipatory licking was increased even when the sound was omitted (Mann-Whitney U-test, p = 4.87E-8), suggesting that mice updated the predicted sound-source position based on their own actions.
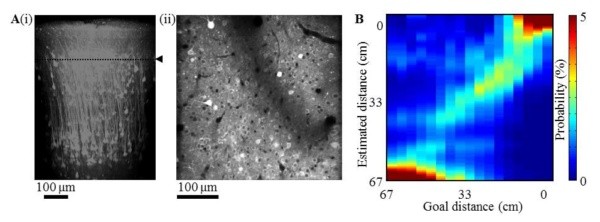
We optically recorded calcium transients of up to 500 neurons simultaneously in each of layers 2 (Fig. Aii), 3 and 5 in 8 mice. A subset of neurons increased the activities as mice approached the sound source (i.e., the goal). This increase of activities was observed both with and without sound inputs in all the layers of PPC (two-sided paired t-test, p = 0.0049 – 2.42E-11), while the increase was observed only during sound inputs in V2 (p = 0.679 – 0.0279).
To test how the increase of activities in PPC and V2 contributed to the prediction of goal distance, we conducted a decoding analysis. Bayesian decoder predicted the goal distance from the recorded population activities (Fig. B): the decoder was trained with the data in continuous condition. In layers 3 and 5 of PPC, the predicted distance significantly decreased both with and without sound inputs (two-sided paired t-test, p = 2.54E-5 and 1.91E-5) consistently with the actual distance to the goal. In contrast, V2 required the sounds for updating distance predictions (p = 0.0752 and 0.0412). These results suggest that PPC, but not V2, realizes action-dependent state prediction in the absence of sensory input.
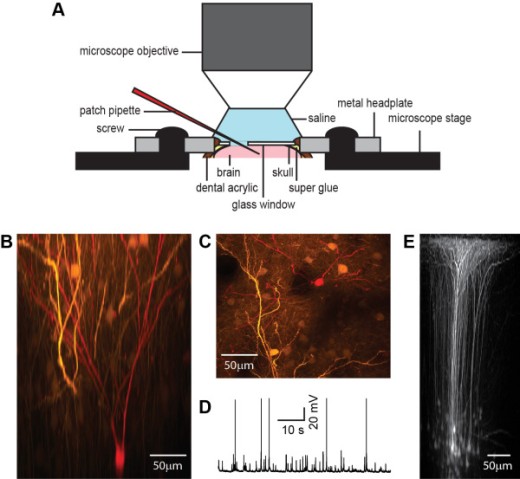
3.3 NMDA Spike/Plateau Potentials in Dendrites of Thalamocortical Neurons
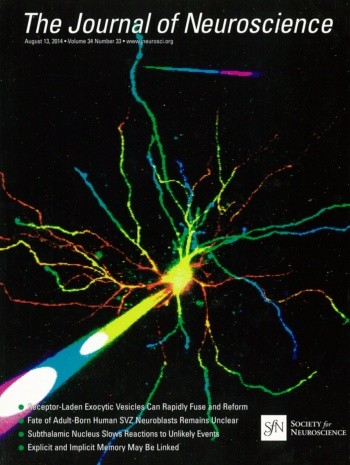
Dendritic NMDA spike/plateau potentials, first discovered in cortical pyramidal neurons, provide supralinear integration of synaptic inputs on thin and distal dendrites, thereby increasing the impact of these inputs on the soma. The more specific functional role of these potentials has been difficult to clarify, partly due to the complex circuitry of cortical neurons. Thalamocortical (TC) neurons in the dorsal lateral geniculate nucleus participate in simpler circuits. They receive their primary afferent input from retina and send their output to visual cortex. Cortex, in turn, regulates this output through massive feedback to distal dendrites of the TC neurons. The TC neurons can operate in two modes related to behavioral states: burst mode prevailing during sleep, when T-type calcium bursts largely disrupt the transfer of signals from retina to cortex, and tonic mode, which provides reliable transfer of retinal signals to cortex during wakefulness. Westudied dendritic potentials inTCneurons with combined two-photon calcium imaging and whole-cell recording of responses to local dendritic glutamate iontophoresis in acute brain slices from mice. We found that NMDA spike/plateaus can be elicited locally at distal dendrites of TC neurons.Wesuggest that these dendritic potentials have important functions in the cortical regulation of thalamocortical transmission. NMDA spike/plateaus can induce shifts in the functional mode from burst to tonic by blockade of T-type calcium conductances. Moreover, in tonic mode, they can facilitate the transfer of retinal signals to cortex by depolarization of TC neurons.
This study was published as the cover story of The Journal of Neuroscience and put into context in a SPIE Newsroom article.
3.4 Multi-Photon Excited Luminescence of Magnetic FePt Core-Shell Nanoparticles
We present magnetic FePt nanoparticles with a hydrophilic, inert, and biocompatible silico-tungsten oxide shell. The particles can be functionalized, optically detected, and optically manipulated. To show the functionalization the fluorescent dye NOPS was bound to the FePt coreshell nanoparticles with propyl-triethoxy-silane linkers and the fluorescence of the labeled particles were observed in ethanol (EtOH). In aqueous dispersion the NOPS fluorescence is quenched making them invisible using 1-photon excitation. However, we observe bright luminescence of labeled and even unlabeled magnetic core-shell nanoparticles with multi-photon excitation. Luminescence can be detected in the near ultraviolet and the full visible spectral range by near infrared multi-photon excitation. For optical manipulation, we were able to drag clusters of particles, and maybe also single particles, by a focused laser beam that acts as optical tweezers by inducing an electric dipole in the insulated metal nanoparticles. In a first application, we show that the luminescence of the core-shell nanoparticles is bright enough for in vivo multi-photon imaging in the mouse neocortex down to cortical layer 5.
This study was published in Biomedical Optics Express.
3.5 Two-Photon Imaging of Ascidians
We image the larvae of transgenic ascidians with two-photon microscopy. This allows us to get higher resolution than with confocal microscopy due to lower scattering artefacts. We studied morphology in fixed larvae and functional signals in vivo.
3.6 Retrograde Labeling of Cortical Neurons
We are exploring the possibilities of retrograde labeling of neurons for functional imaging. We tested different designs of viral vector systems.
3.7 Dendritic Diameters affect the Spatial Variability of Intracellular Calcium Dynamics in Computer Models
The Optical Neuroimaging Unit supported this project of the Computational Neuroscience Unit by experimental data.
There is growing interest in understanding calcium dynamics in dendrites, both experimentally and computationally. Many processes influence these dynamics, but in dendrites there is a strong contribution of morphology because the peak calcium levels are strongly determined by the surface to volume ratio of each branch, which is inversely related to branch diameter. In this study we explore the predicted variance of dendritic calcium concentrations due to local changes in dendrite diameter and how this is affected by the modeling approach used. We investigate this in a model of dendritic calcium spiking in different reconstructions of cerebellar Purkinje cells and in morphological analysis of neocortical and hippocampal pyramidal neurons. We report that many published models neglect diameter-dependent effects on calcium concentration and show how to implement this correctly in the NEURON simulator, both for phenomenological pool based models and for implementations using radial 1D diffusion. More detailed modeling requires simulation of 3D diffusion and we demonstrate that this does not dissipate the local concentration variance due to changes of dendritic diameter. In many cases 1D diffusion of models of calcium buffering give a good approximation provided an increased morphological resolution is implemented.
This project was published in Frontiers in Cellular Neuroscience.
4. Publications
4.1 Journals
- C.J. Roome and B. Kuhn. Chronic cranial window with access port for repeated cellular manipulations, drug application, and electrophysiology. Frontiers in Cellular Neuroscience 8: 379 (2014) .
- S. Augustinaite, B. Kuhn, P.J. Helm, and P. Heggelund. NMDA spike/plateau potentials in dendrites of thalamocortical neurons. Journal of Neuroscience 34(33): 10892-10905 (2014).
- K.M. Seemann and B. Kuhn. Multi-photon excited luminescence of magnetic FePt core-shell nanoparticles. Biomedical Optics Express 5(7): 2446-2457 (2014).
- H. Anwar, C.J. Roome, H. Nedelescu, W. Chen, B. Kuhn, and E. De Schutter. Dendritic diameters affect the spatial variability of intracellular calcium dynamics in computer models. Frontiers in Cellular Neuroscience 8: 168 (2014) .
4.2 Books and other One-Time Publications
Nothing to report
4.3 Oral and Poster Presentations
- O. Jaidar, C.J. Room, Y. Nakano, M. Garcia-Munoz, B. Kuhn, G. Arbuthnott.Thalamocortical axons and deep cortical layer dendritic arbor in motor cortex layer I display a dynamic array of calcium transients during locomotion and systemic blockade of dopamine receptors. BNA 2015 Festival of Neuroscience, Edinburgh, G.B. (2015).
- A. Funamizu, B. Kuhn, K. Doya. Imaging action-dependent state prediction in mouse posterior parietal cortex. FENS-Hertie Winter School, Obergurgl, Austria (2015).
- A. Funamizu, B. Kuhn, K. Doya. Imaging action-dependent state prediction in mouse posterior parietal cortex. Shingakujyutsu meeting, Tokyo, Japan (2014).
- C. Roome, B. Kuhn. Chronic cranial window with access port for repeated cellular manipulations, drug application, and electrophysiology. Society for Neuroscience Meeting, Washington, DC, U.S.A. (2014).
- A. Funamizu, B. Kuhn, K. Doya. Action-dependent state prediction in the parietal cortex of mouse during a virtual navigation task. Society for Neuroscience Meeting, Washington, DC, U.S.A. (2014).
- C. Roome, B. Kuhn. Chronic cranial window with access port for repeated cellular manipulations, drug application, and electrophysiology. Barrels Meeting, Baltimore, MD, U.S.A. (2014).
- S. Augustinaite. NMDA spike/plateau potentials in dendrites of thalamocortical neurons. NIH, Bethesda, MD, U.S.A. (2014).
- A. Funamizu, B. Kuhn, K. Doya. Investigation of action-dependent state prediction in the mouse parietal cortex with two-photon microscopy. Japan Neuroscience Meeting, Yokohama, Japan (2014).
5. Intellectual Property Rights and Other Specific Achievements
Nothing to report
6. Meetings and Events
6.1 Harvard Imaging Symposium on Advanced Light Microscopy
- Date: May 2, 2014
- Venue: Harvard Medical School, Boston, MA, U.S.A.
- Speaker (among others): Bernd Kuhn (OIST)
- Title: Multi-Photon Excited Luminescence of Magnetic FePt Core-Shell Nanoparticles
6.2 Workshop: Okinawa Computational Neuroscience Course 2014
- Date: June 16- July 3, 2014
- Venue: OIST Seaside House
- Organizers: Drs. E. De Schutter, K. Doya, J. Wickens
- Speaker (among others): Bernd Kuhn
- Title 1: Voltage-gated channels and the Hodgkin-Huxley model of neuronal activity
- Title 2: Functional Optical Imaging Methods
6.3 Onna-son/OIST Children’s School of Science 2014
- Date: August 18-22, 2014
- Venue: Fureai Taiken Center
- Speaker (among others): Bernd Kuhn
- Topic: Palaeontology of Okinawa for Grade 1-3
6.4 Barrels Meeting XXVII
- Date: November 13-14, 2014
- Venue: Johns Hopkins University, Baltimore, MD, U.S.A.
- Speakers (among others): Bernd Kuhn (OIST)
- Title: Chronic Cranial Window with Access Port for Repeated Cellular Manipulations, Drug Application, and Electrophysiology
6.5 OIST Neuroscience Day @ Princeton Neuroscience Institute
- Date: November 21, 2014
- Venue: Princeton Neuroscience Institute, Princeton University, Princeton, NJ, U.S.A.
- Speakers:
- Bernd Kuhn (OIST): Multi-photon luminescence of ferromagnetic FePt core-shell nanoparticles
- Christopher J. Roome (OIST): Chronic cranial window with access port for repeated cellular manipulations
- Akihiro Funamizu (OIST): Action-dependent state prediction in mouse posterior parietal cortex
- Sigita Augustinaite (OIST): NMDA spike/plateau potentials in dendrites of thalamocortical neurons
6.6 Vires Acquirit Eundo, Tokyo University - OIST Joint Symposium
- Date: December 12, 2014
- Venue: OIST
- Speakers (among others): Bernd Kuhn (OIST)
- Title: Optical Imaging of Brain Activity
6.7 The 15th RIES-Hokudai International Symposium
- Date:December 16-17, 2014
- Venue: Chateraise Gateaux Kingdom, Sapporo, Japan
- Speakers (among others): Bernd Kuhn (OIST)
- Title: Two-Photon Microscopy - A Tool For Many Different Applications
6.8 CSN School on Neurotechniques 2015
- Date: March 23-27, 2015
- Venue: University of Padua, Padua, Italy
- Speakers (among others): Bernd Kuhn (OIST)
- Title 1: In Vivo Imaging
- Title 2: Solvatochromism and Electrochromism
6.9 Planning of ICIIBMS 2015
- First International Conference on Intelligent Informatics and BioMedical Sciences
- Date: November 28-30, 2015
- Venue: OIST
- Organized by researchers of the University of the Ryukyus, the Okinawa National College of Technology, and OIST